1. Carbon (C)
Diamonds and graphite have long been known and naturally occur in the Earth's crust. Graphite, also known as grafit in Greek, meaning to write.
Carbon has two interesting stories:
Why choose carbon-12 isotope in the atomic mass unit (AMU) scale of chemical elements (CE)?
In 1803, hydrogen, being the lightest element, was chosen as the AMU unit. However, since most CEs easily form compounds with oxygen as oxides, the atomic masses had to be compared to the atomic mass of oxygen. Therefore, 1/16 of the oxygen atom was chosen as the unit of measurement and called the oxygen unit.
With scientific advancements, a contradiction arose. In the early 20th century, natural oxygen was determined to be a mixture of isotopes. Scientists still considered the oxygen unit as 1/16 of natural oxygen (meaning all isotopes of oxygen), but for atomic physics, this unit was inaccurate. Physicists recognized the oxygen unit as 1/16 of the oxygen-16 isotope.
The measurement unit with two scales is the physical scale and the chemical scale. To resolve the contradiction, the International Conference decided to switch to carbon in 1961. The advantage is that natural carbon only has 2 stable isotopes, C-12 and C-13, with C-12 accounting for 98.892% of all carbon atoms.
So, since 1961, scientists have unanimously chosen the atomic mass unit as 1/12 of the C-12 atom, denoted as amuC.
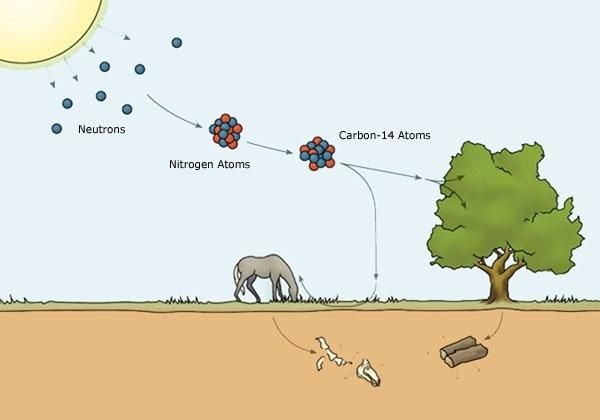
Using C-14 isotope to determine the age of archaeological plant remains
Cosmic rays are dangerous due to neutron emissions. Thanks to the high-altitude ozone layer, it blocks cosmic rays from reaching the Earth's surface. Cosmic neutron particles interacting with N-14 isotopes create the C-14 isotope. C-14 is not stable and undergoes decay in nature with a half-life period T=5570 years.
Due to photosynthesis, plants absorb CO2 in the atmosphere for growth. CO2 in the atmosphere contains a tiny portion of 14CO2 (for every 1 million normal CO2 molecules, there is 1 molecule of 14CO2).
Because the C-14 production reaction in the atmosphere is continuous, when the plant is still alive, the C-14 content remains almost unchanged. When the plant dies and photosynthesis no longer occurs, the C-14 amount gradually decreases according to the above-mentioned decay cycle.
If you take a piece of wood of the same type and size, but freshly cut compared to a piece of wood from clothing in ancient tombs, you can immediately determine how much C-14 remains. And we can quickly find the age of the tomb.