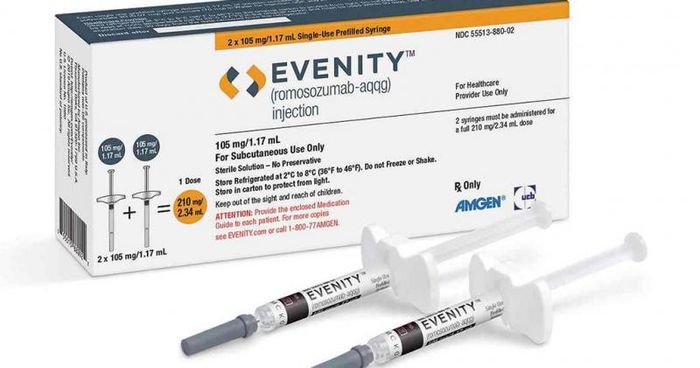
1. Dual-Action Bone Density Medication
Osteoporosis, a condition where bones become weak and prone to fracture, has seen significant advancements in treatment. Since 1995, medications primarily focused on preventing bone erosion, slowing bone loss. In 2002, drugs promoting bone formation became popular, stimulating bone-building cells. However, these drugs also increased bone resorption, reducing some of their reinforcing effects. A new medication approved by the FDA in April 2019, romosozumab (marketed as Evenity by AMGEN), addresses this drawback. Administered via injection twice a month for a year, this breakthrough in osteoporosis treatment offers efficacy not only in bone health but also in the morale of patients and healthcare providers.
2. Treatment for Transthyretin Amyloid Cardiomyopathy
Transthyretin amyloid cardiomyopathy (ATTR-CM) is a rare, progressive, challenging to diagnose condition associated with high mortality rates and previously lacked successful treatment. Typically, ATTR-CM is diagnosed only after symptoms become severe, with patients surviving an average of 2 to 3.5 years due to previous treatments limited to heart transplantation. A new medication, tafamidis (marketed as Vyndaquel by Pfizer), stabilizes Transthyretin, slowing amyloid formation, significantly reducing amyloid deposition in the heart, thereby showing a notable reduction in mortality risk. May 2019 marked FDA approval for tafamidis, the first drug used to treat this increasingly recognized condition. Phase 3 clinical trial results demonstrate tafamidis reduces mortality risk by 30% in patients receiving this breakthrough therapy.
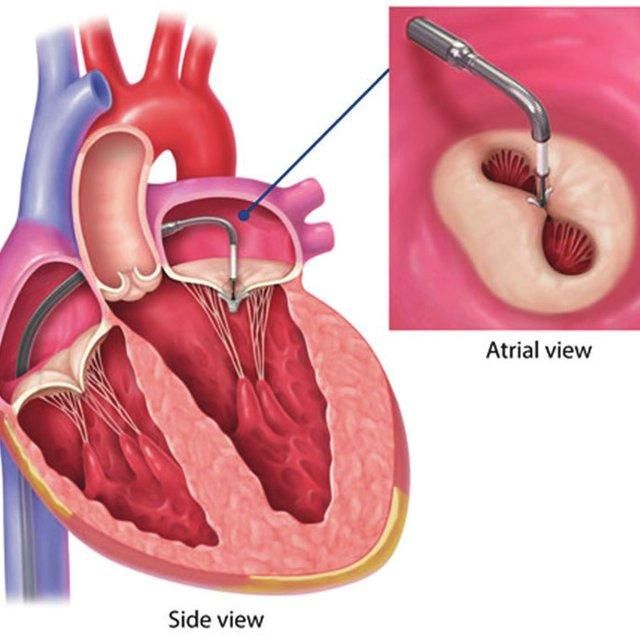
3. Advancement in Minimally Invasive Techniques for Mitral Valve Repair via Transcatheter Approach (MitraClip)
Mitral valve regurgitation is relatively common, estimated at a rate of 1 in 10 people over 75 years old. Previously, treatment involved high-risk open-heart surgery with extended recovery periods lasting several months. In March 2019, the FDA expanded approval of this minimally invasive technique to patients with secondary mitral valve regurgitation or mitral valve regurgitation due to left ventricular enlargement. The expanded indication for patients with mitral valve regurgitation who showed no signs of symptomatic improvement after undergoing other treatments provides an important new treatment option for physicians and patients by eliminating the fear and barriers associated with heart surgery, reducing treatment risks.
4. Closed-loop Spinal Cord Stimulation in Chronic Pain Treatment
Chronic pain is a debilitating condition characterized by recurring episodes of prolonged pain lasting over 3 months. Patients with chronic pain experience psychological disorders such as anxiety, depression, mood changes affecting daily social activities.
Closed-loop spinal cord stimulation allows the implanted device to interact better with the spinal cord, enabling real-time communication with spinal nerve cells. This is the first system capable of measuring nerve responses to stimulate and adjust all electrical impulses based on real activities. Closed-loop spinal cord stimulation is now indicated for treatment in European countries, providing relief for chronic pain patients everywhere.
5. Therapy for Peanut Allergy
Peanut allergy is a common cause of severe allergic reactions with symptoms that can be life-threatening, such as anaphylaxis. Currently, peanut allergies are increasing in children. Recently, the development of a new oral immunotherapy product to gradually build tolerance to peanuts has provided an opportunity for patients to combat peanut allergies.
In 2019, an FDA expert panel recommended approval of the first therapy for peanut allergy. It is a capsule formulation containing very small amounts of peanut protein. Over time, the dose is increased, and children develop a tolerance to peanuts.

6. Antibiotic Coating to Prevent Infections of Cardiac Assist Devices
Globally, about 1.5 million patients undergo surgery to implant these cardiac assist electronic devices each year. Infection is a dangerous complication during the implantation process of these devices. So far, using oral antibiotics just before surgery has been the best and only proven way to prevent infection. However, infections continue to occur.
The recent development of an antibiotic coating product helps ensure slow release of two types of antibiotics at the implant site for seven days after surgery. Slow release of antibiotics over a week will minimize the risk of infection. Initial findings from this trial, published in March 2019, show a 40% reduction in major infection cases, providing greater safety for patients undergoing cardiac assist device implantation procedures.
7. Biotechnology in Orthopedic Surgery
Biotechnology – including cellular components, blood components, growth factors, and other natural substances – harnesses the body's own power to promote healing and reduce inflammation in treatment. These factors are changing the landscape of orthopedic surgery, allowing for rapidly improved treatment outcomes.
This new biotechnology technique uses an injected foam containing autologous biological factors combined with the patient's own blood to stimulate healing of anterior cruciate ligaments, preserving tissue rather than cutting it away. Biotechnology holds great promise in providing every orthopedic surgery patient with a faster, more natural, and effective recovery.

8. PARP Inhibitors - Maintenance Therapy in Ovarian Cancer
Ovarian cancer is a malignant tumor originating from one or both ovaries. Cancer cells are abnormal cells that grow uncontrollably and do not follow the body's normal needs. These cancer cells can invade and destroy surrounding tissues and organs. Moreover, they can metastasize to distant organs in the body, causing secondary cancer at those sites.
PARP (Poly ADP ribose polymerase) is an enzyme involved in DNA repair and prevents cell self-destruction in the body, or in other words, PARP is a protein that tumors rely on to repair defects. When the body has cancer, tumor cells hijack this function of PARP and exploit it to help the tumor grow. Inhibiting PARP activity to prevent DNA repair and enhance cell death has emerged as a promising therapy in cancer treatment and has been brought into clinical trials.
9. Acid Bempedoic Aids in Lowering Cholesterol in Statin Intolerant Patients
Cholesterol is a pale yellow sterol lipid, transported in the blood plasma and found in the cell membranes of body tissues. High cholesterol is a condition where the cholesterol levels in the blood are higher than normal. When cholesterol levels are elevated, the body will deposit LDL cholesterol in the blood vessels. These deposits can obstruct the flow of blood through the arteries. Consequently, the heart may not receive enough oxygen-rich blood, leading to angina. Reduced blood flow to the brain can also result in a stroke.
Acid bempedoic works by inhibiting a key enzyme used by the body to produce cholesterol, particularly not accumulating in muscles, reducing the risk of muscle pain. If FDA approved, bempedoic acid could be an effective and suitable adjunctive therapy in the goal of reducing cholesterol for patients.

