Mol concentration represents the relationship between the number of moles of a solute and the volume of the solution. To calculate mol concentration, you can begin with the number of moles and volume, mass and volume, or moles and milliliters (ml). Using these variables, apply the basic mol concentration formula to get an accurate result.
Steps
Calculate Mol Concentration from Moles and Volume

It's important to know the basic formula for calculating mol concentration. Mol concentration is the number of moles of a solute divided by the volume of the solution in liters. Thus, the formula is: Mol concentration = moles of solute / liters of solution
- Example: What is the mol concentration of a solution containing 0.75 mol NaCl in 4.2 liters of solution?

Analyzing the Problem. To calculate molar concentration, you need the number of moles and the volume of the solution in liters. You do not need to compute these values as they are already provided in the question.
- Example:
- Number of moles = 0.75 mol NaCl
- Volume = 4.2 L

Divide the number of moles by the volume. The result of dividing the number of moles by the volume gives you the molar concentration, which is the number of moles per liter of solution.
- Example: molar concentration = number of moles / liters of solution = 0.75 mol / 4.2 L = 0.17857142

Record the result. Round the result to two or three decimal places, depending on the teacher's instructions or the problem's requirements. When noting the result, abbreviate 'molar concentration' as 'M' and include the chemical symbol of the solute.
- Example: 0.179 M NaCl
Calculating Molar Concentration from Mass and Volume

Know the basic formula for calculating molar concentration. Molar concentration shows the relationship between the number of moles of a solute and the volume of the solution. The formula for molar concentration is as follows: molar concentration = moles of solute / liters of solution
- Example question: Calculate the number of moles in a solution made by dissolving 3.4 g of KMnO4 in 5.2 liters of water.

Analyze the Problem: To find molar concentration, you need the number of moles and the volume of the solution in liters. If these values are not provided but you know the volume and mass of the solution, you can first determine the number of moles before calculating the molar concentration.
- Example:
- Mass = 3.4 g KMnO4
- Volume = 5.2 L
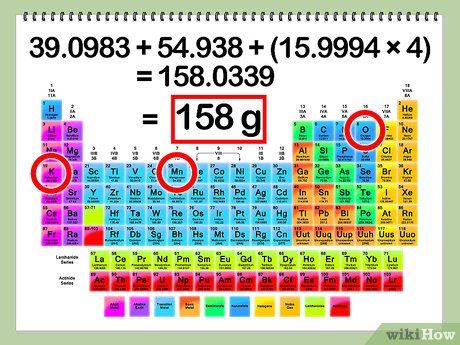
Calculate the Molar Mass of the Solute. To calculate the number of moles of the solute from the mass or grams of the solute, you first need to determine the molar mass of the solute. The molar mass can be found by adding up the atomic masses of each element present in the solution. To find the atomic mass of each element, use the periodic table of elements.
- Example:
- Atomic mass of K = 39.1 g
- Atomic mass of Mn = 54.9 g
- Atomic mass of O = 16.0 g
- Total atomic mass = K + Mn + O + O + O + O = 39.1 + 54.9 + 16 + 16 + 16 + 16 = 158.0 g

Convert Grams to Moles. Once you have the molar mass, you need to multiply the mass of the solute in the solution by the conversion factor of 1 mole per molar mass of the solute. This calculation will give you the number of moles of the solute.
- Example: mass of solute * (1 / molar mass of the solute) = 3.4 g * (1 mol / 158 g) = 0.0215 mol

Divide Moles by Liters. After calculating the number of moles, divide this value by the volume of the solution in liters to obtain the molar concentration of the solution.
- Example: molar concentration = number of moles of solute / number of liters of solution = 0.0215 mol / 5.2 L = 0.004134615

Record the Result. You need to round the result according to the teacher's instructions, typically to two or three decimal places. Additionally, when writing the result, abbreviate 'molar concentration' as 'M' and include the chemical symbol of the solute.
- Example: 0.004 M KMnO4
Tính Nồng độ Mol từ Số mol và Mililít dung dịch

Cần nắm công thức tính nồng độ mol. Để tính nồng độ mol. Bạn cần tính số mol của chất tan trong một lít dung dịch chứ không phải mililít dung dịch. Công thức chung để tính nồng độ mol là: nồng độ mol = số mol chất tan / số lít dung dịch
- Ví dụ: Tính nồng độ mol của một dung dịch chứa 1,2 mol CaCl2 trong 2905 mililít nước.

Phân tích đề bài. Để tính nồng độ mol, bạn cần có số mol chất tan và thể tích dung dịch tính theo lít. Nếu thể tích dung dịch nêu trong đề bài được tính theo mililít, bạn cần chuyển sang thể tích tương đương theo lít trước khi tính toán.
- Ví dụ:
- Số mol = 1,2 mol CaCl2
- Thể tích = 2905 ml

Đổi mililít sang lít. Để đổi dung dịch từ đơn vị là mililít sang lít, bạn chia số mililít cho 1000, vì mỗi lít tương đương với 1000 mililít. Bạn cũng có thể chuyển đổi từ mililít sang lít bằng cách dịch chuyển dấu phẩy thập phân sang trái 3 chữ số.
- Ví dụ : 2905 ml * (1 L / 1000 ml) = 2,905 L

Chia số mol cho số lít. Sau khi có số lít, bạn có thể tính được nồng độ mol bằng cách lấy số mol chia cho số lít dung dịch.
- Ví dụ: nồng độ mol = số mol chất tan / số lít dung dịch = 1,2 mol CaCl2 / 2,905 L =0,413080895

Record the result. Remember to round the result to two or three decimal places, or as instructed by your teacher. When writing the result, abbreviate 'molar concentration' as 'M' followed by the chemical symbol of the solute.
- Example: 0.413 M CaCl2
Additional Practice

Calculate the molar concentration of a solution by dissolving 5.2 g NaCl into 800 ml of water. Identify the values given in the problem: mass in grams and volume in milliliters.
-
- Mass = 5.2 g NaCl
- Volume = 800 ml water
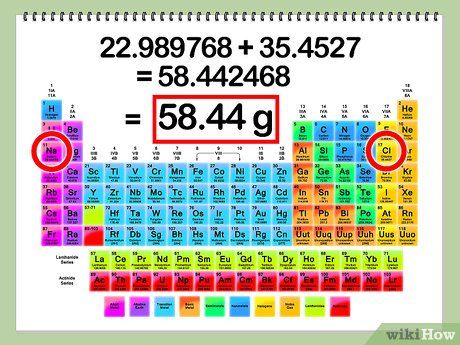
Find the molar mass of NaCl by adding the atomic masses of Na and Cl.
- Atomic mass of Na = 22.99 g
- Atomic mass of Cl = 35.45 g
- Molar mass of NaCl = 22.99 + 35.45 = 58.44 g

Multiply the mass of the solute by the conversion factor to obtain moles. In this example, the molar mass of NaCl is 58.44 g, so the conversion factor is '1 mole / 58.44 g'.
- Number of moles of NaCl = 5.2 g NaCl * (1 mol / 58.44 g) = 0.0898 mol ≈ 0.09 mol
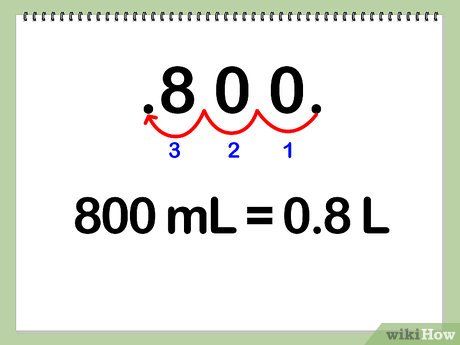
Convert 800 ml of water to liters.
- You can multiply 800 ml by the conversion factor from milliliters to liters, which is 1 L / 1000 ml.
- To simplify this process, you can move the decimal point three places to the left.
- Volume = 800 ml * (1 L / 1000 ml) = 800 ml / 1000 ml = 0.8 L

Divide the moles of solute by the volume of solution in liters. To calculate molar concentration, divide 0.09 moles of solute (in this case, NaCl) by the volume of the solution in liters.
- Molar concentration = number of moles of solute / number of liters of solution = 0.09 mol / 0.8 L = 0.1125 mol/L

Record the final result. Round the result to two or three decimal places and abbreviate 'molar concentration' as 'M' followed by the chemical symbol of the solute.
- Result: 0.11 M NaCl
