The average atomic mass is not the direct measurement of a single atom's mass. Instead, it represents the average mass of atoms calculated from a representative sample of the element. If it were possible to measure the mass of billions of individual atoms, you could compute this value by averaging them. A more practical approach is based on data from the different isotopes of the chemical element.
Steps
Calculate the Average Atomic Mass
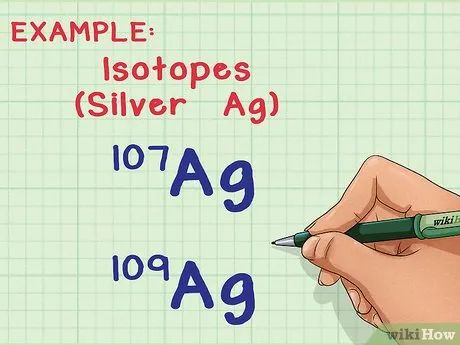
Understand Isotopes and Atomic Mass. In nature, most elements exist in multiple forms, or isotopes. The only difference between two isotopes of the same element is the number of neutrons in the atom, and the number of neutrons affects the atomic mass. The calculation of the average atomic mass takes this variation into account, providing the average mass per atom in a sample containing many atoms.
- For example, the element silver (Ag) has two isotopes in nature: Ag-107 and Ag-109 (or 107Ag and 109Ag). Isotopes are named based on their “mass number,” the total of protons and neutrons in an atom. This means Ag-109 has two more neutrons than Ag-107, making its atoms slightly heavier.

Determine the mass of each isotope. You will need two pieces of information for each isotope, which can be found in reference books or online resources such as the website webelements.com. The first is the atomic mass or the atomic weight of each isotope. Isotopes with more neutrons will have a higher mass.
- For example, the isotope Ag-107 has an atomic mass of 106.90509 amu (atomic mass unit). The isotope Ag-109 is slightly heavier with a mass of 108.90470.
- The decimal numbers at the end might slightly vary in different references. Do not include any number in parentheses following the mass.

Write the natural abundance of each isotope. This refers to the relative frequency of each isotope, expressed as a percentage of the total number of atoms of that element. This information can be found in the same sources that list the atomic masses. The total natural abundance of all isotopes should add up to 100% (although small rounding errors may occur).
- The isotope Ag-107 has an abundance of 51.86%. The isotope Ag-109 is less common, with an abundance of 48.14%. This means a typical silver sample contains 51.86% Ag-107 and 48.14% Ag-109.
- Ignore any isotopes that do not have this natural abundance value. These isotopes do not naturally occur on Earth.

Convert the isotope abundances to decimal form. Dividing these percentages by 100 will give the corresponding decimal values.
- For the silver sample mentioned earlier, the isotope abundances are 51.86 / 100 = 0.5186 and 48.14 / 100 = 0.4814.

Calculate the average atomic mass. The average atomic mass of an element with n isotopes can be found using the formula: (atomic massisotope 1 * abundanceisotope 1) + (atomic massisotope 2 * abundanceisotope 2) + ... + (atomic massisotope n * abundanceisotope n. This is an example of a “weighted average,” meaning that the greater the abundance of an isotope, the more influence it has on the final result. Here's how to apply this formula to silver:
- Average atomic massAg = (atomic massAg-107 * abundanceAg-107) + (atomic massAg-109 * abundanceAg-109)
=(106.90509 * 0.5186) + (108.90470 * 0.4814)
= 55.4410 + 52.4267
= 107.8677 amu. - Check the periodic table to verify the result. The average atomic mass is always listed below the element's symbol.
Use the result
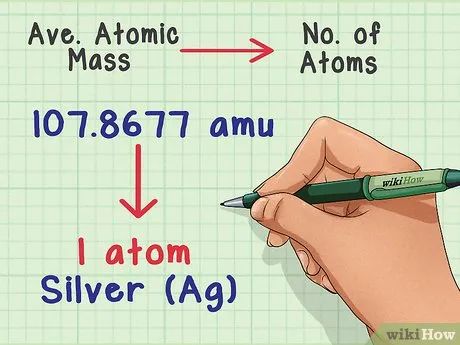
Convert mass to atomic number. The average atomic mass reveals the relationship between mass and atomic number in a representative sample of the element. This is very useful in chemistry labs, as it is nearly impossible to count individual atoms precisely, but mass can be easily determined. For instance, you can weigh a silver sample and know that for every 107.8677 amu, there will be one silver atom.

Convert to molar mass. Atomic mass units are very small, so chemists often use grams for mass. Fortunately, we have definitions for these concepts, so the conversion is straightforward. Simply multiply the average atomic mass by 1 g/mol (the molar mass constant) to get the result in g/mol. For example, 107.8677 grams of silver contains one mole of silver atoms.

Calculate the average molecular mass. Since a molecule is made up of atoms, you can sum the masses of all the atoms to find the molecular mass. If you use the average atomic mass (instead of the mass of a specific isotope), the result will be the average molecular mass of a natural sample. Here’s an example:
- A water molecule with the chemical formula H2O contains two hydrogen (H) atoms and one oxygen (O) atom.
- Hydrogen has an average atomic mass of 1.00794 amu, while oxygen has an average atomic mass of 15.9994 amu.
- Thus, the average molecular mass of H2O is (1.00794)(2) + 15.9994 = 18.01528 amu, equivalent to 18.01528 g/mol.
Tips
- The term relative atomic mass is sometimes used interchangeably with average atomic mass. There is a slight difference because relative atomic mass is unitless; it is a mass measurement relative to the carbon-12 atom. As long as you use atomic mass units in average atomic mass calculations, both values are essentially the same.
- The number in parentheses after an atomic mass indicates the margin of error. For example, an atomic mass of 1.0173 (4) means the normal atomic mass of that element falls within a range of 1.0173 ± 0.0004. You do not need to consider this number unless specifically instructed to do so.
- On the periodic table, the average atomic mass of an element will generally be higher than that of the element preceding it, with a few exceptions. This is a quick way to check your results.
- 1 atomic mass unit equals 1/12 of the mass of a carbon-12 atom.
- The isotopic abundances are calculated based on the typical natural sample on Earth. Elements that do not naturally occur, such as meteors or substances created in labs, may have different isotopic abundances, and therefore their average atomic masses can differ.
Warning
- Atomic mass is always written in atomic mass units (amu or u), sometimes called daltons (Da). Never write a different mass unit (such as kilograms) after this number without converting it.
What You Need
- Pencil
- Paper
- Calculator
- Data on the natural isotopic abundance.
- Data on the atomic mass units for isotopes.
