In chemistry, pH is the scale that measures the acidity or alkalinity of a substance. The pH scale ranges from 0 to 14, with values near 0 being highly acidic, values near 14 being strongly alkaline, and 7 being perfectly neutral. In gardening and agriculture, soil pH can have a significant impact on plant health and growth. While most plants thrive in a pH range of 6.0 to 7.5, some plants grow best in a narrower pH range, so gardeners should learn the basics of pH control. Read step 1 below to begin learning how to lower your soil's pH.
Steps
Conduct a pH Test

Test your soil's pH. Before adding anything to alter your soil’s pH, you should always first test it to determine how much you need to lower the pH. You can purchase a testing kit from a garden center or take a soil sample to a local rural development office for testing.
Dig 5 small holes in the planting area. The pH level of garden soil is fairly easy to determine using a pH testing kit, which are commonly available at home goods or garden supply stores at an affordable price. To begin testing, you need to collect soil samples from the area you want to test. Dig 5 small holes (about 15-20 cm deep). Choose random spots within the garden area to get an accurate "average" pH level of the soil. There's no need to keep the soil you remove from these holes.
- Note that the instructions in this section are general; be sure to follow the specific instructions that come with your pH test kit.

Collect soil samples from each hole. Next, use a spade or trowel to cut a "slice" of soil from the side of each hole. This slice should be crescent-shaped and about 1.5 cm thick. Try to take soil samples of equal size. Place all the soil samples in a clean, dry bucket.
- Ensure that the total amount of soil you collect is at least 1 liter or more. For most testing methods, this will be a sufficient amount.
Mix the soil in the bucket and spread it on newspaper to dry. Allow the soil to dry until it no longer feels moist.
- It's crucial that the soil is completely dry before testing the pH, as moisture can skew the results.

Use the test kit to accurately determine the pH level of the soil. The testing method may vary depending on the type of kit you're using. For many common test kits, you'll place a small amount of soil into a test tube, add a few drops of the testing solution, shake well, and allow the mixture to settle for a few hours. Finally, the color of the solution will change; by comparing the color of the solution to the chart provided with the kit, you can determine the pH level of the soil.
- There are many different types of test kits, so be sure to read the instructions that come with your kit. For example, some newer electronic pH testers provide an almost instant reading of the soil's pH using a metal probe. You can also collect a soil sample and bring it to a rural development office for free testing.
Use methods to lower the pH level
Adding Organic Matter. Various organic materials such as compost, manure, and acidic mulching materials (for example, pine needles) can gradually reduce the soil's pH. As organic matter decomposes, bacteria and microorganisms break it down, releasing acidic byproducts. Since organic matter takes time to decompose and alter soil pH, this method is best suited for long-term goals but may not yield significant results in the short term. Many gardeners choose to add organic matter annually to steadily lower the soil's pH. However, this method is not as effective on heavy clay soils.
- In addition to reducing pH, organic matter can also improve soil structure, particularly by enhancing drainage and aeration.
Adding Aluminum Sulfate. For a quicker pH reduction, it is better not to rely on slowly decomposing organic materials. Instead, use one of the many acidifying additives available at garden centers. Among these, aluminum sulfate is one of the fastest acting options. It lowers soil pH immediately upon dissolving, providing an instant effect, making it ideal for rapid soil pH adjustment in gardening. Thus, aluminum sulfate is an excellent choice for quickly reducing soil pH.
- The required amount of aluminum sulfate varies depending on the initial pH level. Generally, for a 30 m2 area, you'll need 0.5 kg of aluminum sulfate to reduce the pH by 1 unit (e.g., from 7 to 6, or 6 to 5). However, excessive use of this additive can harm plants, so it’s recommended to consult reliable online resources (such as here) for accurate application details.
- Avoid using aluminum sulfate over large areas, as it can lead to aluminum accumulation and toxicity in the soil.
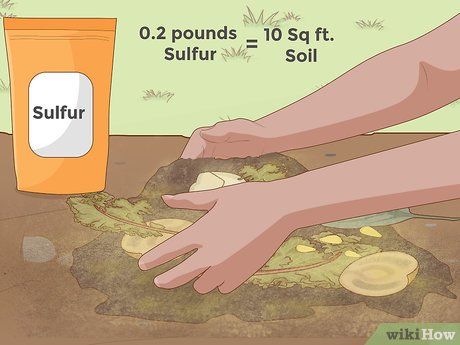
Adding Sulfur. Another useful additive for reducing soil pH is sublimed sulfur. Compared to aluminum sulfate, sulfur is usually cheaper, more potent (in terms of the quantity needed), and works slower. This is because sulfur needs to be processed by soil bacteria to form sulfuric acid, a process that takes time. Depending on soil moisture, bacterial levels, and temperature, sulfur may take several months to show a noticeable effect on soil pH.
- As mentioned above, sulfur is generally used in smaller amounts than aluminum sulfate to achieve the same pH reduction. Typically, around 90 g of sulfur is needed to decrease the pH by 1 unit over a 30 m2 area. You can check online resources (like here) for further guidance on its use.
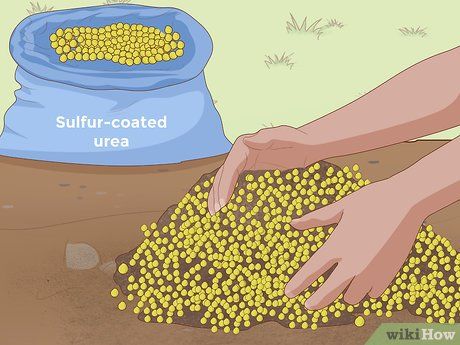
Adding Urea-Sulfur Coated Fertilizer. Like sulfur and aluminum sulfate, urea-sulfur coated fertilizers can gradually increase soil acidity (lower pH). As a fertilizer, urea acts quickly, showing results within one or two weeks after application. Urea-sulfur coated fertilizers are commonly found in many types of fertilizers, so if you are planning to fertilize your plants, you may not need to look for a separate acidifying additive. You can simply choose a fertilizer that contains urea-sulfur.
- The urea-sulfur content varies across different fertilizers, so be sure to read the instructions on the packaging to determine the right dosage for your garden’s needs.
- Urea-sulfur is a slow-release fertilizer, providing nutrients gradually as plants require them, instead of delivering a one-time dose.
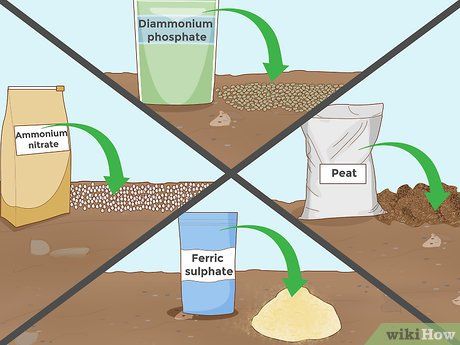
Adding Other Acidifying Additives. In addition to the above additives, several other substances can help reduce soil pH. Many of these are commonly found in fertilizer blends, while others are available as pure additives. The time required and the quantity needed can vary widely depending on the specific substance, so it’s important to read the packaging or consult with an experienced staff member at your garden supply store. Some other acidifying additives include:
- Diammonium phosphate
- Iron sulfate
- Peat moss
- Ammonium nitrate
Plant species that tolerate alkaline soil. If the soil has a high alkalinity level, which is unsuitable for acid-loving plants, planting species that thrive in alkaline conditions can gradually lower the soil's pH throughout the plant's life cycle. As the plant grows, matures, and decays, the organic matter it returns to the soil supports bacterial growth and helps decrease the soil's pH (similar to adding organic material like mulch or fertilizers). This method is one of the slowest ways to reduce soil pH, as the plant must develop before organic matter starts being returned to the soil. Some alkaline-tolerant plants include:
- Evergreen shrubs (such as golden yew, California lilac)
- Deciduous shrubs (like lilac, orange jasmine, and forsythia)
- Perennial plants (such as sedum, rockrose)
Identify plants that prefer low pH soil.
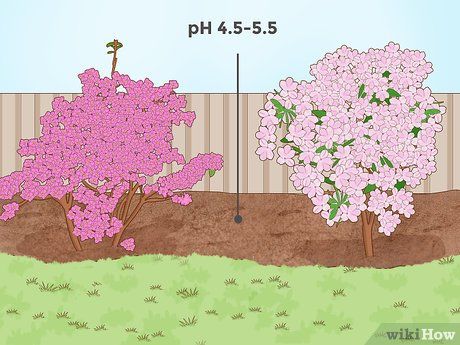
Lowering soil pH for shrubs like azaleas. Some flowering shrubs, such as azaleas, require slightly acidic soil to thrive. These plants are often native to regions with high rainfall, such as the Pacific Northwest of the United States (where rainfall helps reduce soil pH). For these shrubs, an optimal pH range is 4.5–5.5, although a pH up to 6 can still be acceptable.
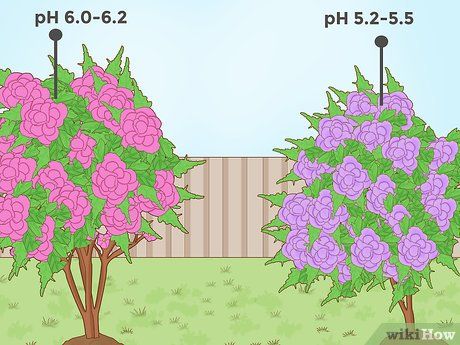
Reducing pH for flowering plants like begonias and hydrangeas. Many colorful flowers, such as petunias and begonias, grow best in slightly acidic soils. For some of these flowers, adjusting the pH from
mildly to
strongly acidic can significantly alter the flower color. For example, hydrangeas grown in soil with a pH around 6.0–6.2 will produce pink flowers, but if the pH is lowered to around 5.2–5.5, the flowers will turn purple/blue.
- The blue color of hydrangea flowers in low pH soil is due to the presence of aluminum ions. When the soil's pH is low, the plant can absorb aluminum more easily, which results in the blue color of the petals.
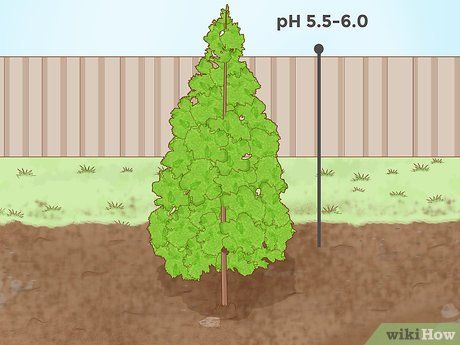
Lowering soil pH for evergreen plants. Many coniferous evergreen trees grow best in mildly acidic soil. For example, spruces, firs, and pines thrive in soil with a pH of around 5.5–6.0. Additionally, the leaves of these trees can contribute neutral or alkaline organic matter that helps reduce soil pH as they decompose.
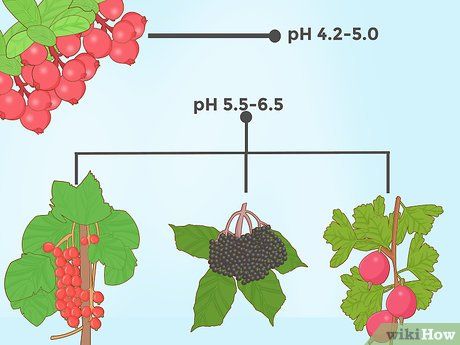
Lowering soil pH for certain berry plants. One of the most acid-loving plants is the blueberry. It thrives in highly acidic soil (a pH between 4 and 5 is ideal). However, several other berry plants also prefer acidic conditions. For instance, cranberry plants grow best in soil with a pH of 4.2–5.0, while other varieties like elderberries, Greek grapes, and huckleberries flourish in soil with a pH range of 5.5–6.5.
Lowering pH slightly below neutral for ferns. Most ferns cultivated in gardens prefer soil with a pH under 7; even those that tolerate alkaline soil can withstand mildly acidic conditions. For example, fox tail ferns prefer a pH around 7–8 but can survive in soil with a pH of 6. Some fern species can even tolerate soil with a pH as low as 4.
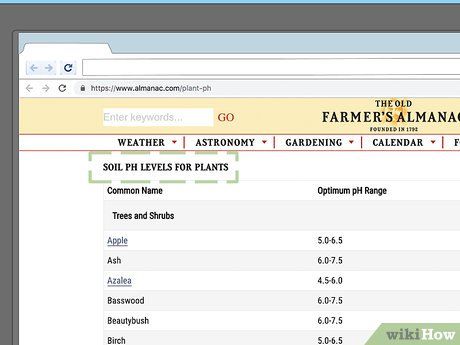
Refer to gardening guides for a complete list of acid-loving plants. There is such a vast number of plants that can thrive or grow well in low pH soil that it's impossible to list them all in this article. For more information, it's best to consult comprehensive plant resources. You can typically find these materials at garden supply stores or specialty bookstores, though many guides are also available online. For example,
The Old Farmer's Almanac has a list of pH preferences for various plants, which you can access
here.
Advice
- Some soil amendments come in spray form.
- It is important not to overuse any soil amendments, as excessive application can have long-term negative effects on both the soil and the environment.
- Plants growing in soil with an unsuitable pH will not grow well because some nutrients become bound in the soil, making them unavailable to the plants.
- The effect of elemental sulfur can last through several seasons.
- Elemental sulfur is most effective when applied during the spring months and is much less effective after the plants are already planted.
- Soil pH affects everything from water drainage to soil erosion.
- Use natural compost whenever possible, as it will benefit the plants by adding nutrients. Composting is an excellent way to recycle grass clippings and kitchen waste.
- Elemental sulfur and compost promote biological reactions, while aluminum sulfate and iron sulfate trigger chemical reactions.
Warning
- Excessive use of aluminum sulfate can lead to soil contamination.
- If urea, aluminum sulfate, or sulfur is accidentally spilled on plant leaves, it is essential to wash the leaves thoroughly with clean water. Chemicals left on the leaves can cause "burn" damage and harm the plant.

















