Nickel plating involves applying a layer of nickel or a nickel alloy to a metal object. If you're looking to do this at home, the only feasible option is electroplating, though other methods are available commercially. There are two main reasons for nickel plating: to protect the underlying material or to enhance the appearance of the plated item. Whether you want to rejuvenate an old piece of jewelry or protect a bolt on your bike, read on to learn more about the nickel plating process.
Steps
Can you nickel plate at home?

Yes, but you must wear gloves and safety goggles. All you need are two nickel electrodes, which you can order online. You’ll also need some white vinegar and a few spring clamps to connect to a battery or power source. Make sure the room is well-ventilated by opening a window and turning on a fan. Don protective gloves, a face mask, and safety goggles.
- This process is called electroplating, and it’s the only method that allows you to nickel plate at home. While there are more efficient methods available, they require professional assistance.
How to set up a nickel plating system?

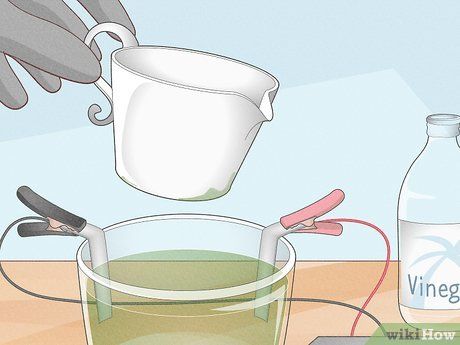
Pour vinegar and a bit of salt into a glass bowl. Hang the two nickel pieces over the edge of the bowl so they are half submerged in the vinegar. Take the two ends of the power source cables and attach the positive and negative clips to the two nickel pieces. Clip the positive wire to one nickel piece and the negative wire to the other. Turn on the power switch and wait.
- For the power source, you can use a 6V or 12V battery. Alternatively, you can buy a dedicated plating power supply with built-in spring clips. If you want to make use of available materials, you can use an old phone charger, cut the cable, separate the two wires, and connect them to the nickel pieces.
- As long as the output current is no more than 1 amp, the process will be safe.
How to nickel plate an object?

Dip the object into the solution when the vinegar turns green. The salt, vinegar, and nickel electrodes will create an electrolyte in the solution, causing it to turn green. When this happens, the solution is charged. Clean the metal object you want to plate, then disconnect the power. Leave the positive nickel electrode over the edge of the bowl but remove the negative electrode. Suspend the object on a copper wire and dip it into the solution.
- The nickel plating process takes about 20 minutes to complete.
- Suspend the object in the solution to prevent it from sinking to the bottom of the bowl. If the object sinks, it won’t be plated evenly on all sides.
Does the nickel plating wear off?

The plating will wear off over time. However, this process takes a long time, by which point the object will have already been heavily worn. There are two primary benefits to nickel plating an item. First, it enhances the object’s appearance, and second, it helps protect the material underneath from damage. While the nickel coating will eventually dull, this takes quite a long time, and it’s a sign that the plating is effectively performing its protective function for the surface beneath.
Is nickel plating expensive?

This depends on the object being plated and the plating method used. There are various nickel plating methods, and plating computer parts requires much more precision than plating a simple bolt. Luckily, plating companies will usually offer free quotes. Contact plating businesses to inquire about their pricing based on the object you wish to plate.
- If you want to plate the item at home, you’ll need two nickel electrodes. You will also need to buy two spring clips, but the cost is minimal.
How can you tell if something is nickel-plated?

Scratch the object and soak it in salt water for 24 hours. A nickel-plated object will corrode in the saltwater, and the color will noticeably change. Pure nickel will not corrode or discolor.
- If you don’t want to risk damaging the item, take it to a jeweler for an evaluation. They will be able to determine if the item is made of pure nickel.
What color is nickel plating?
If you plate at home, it will have a silver color, similar to regular nickel. If the plating process is not performed perfectly, the nickel may have a pale yellow hue. The plating method affects the surface texture, not the color. Home nickel plating results in a shiny surface, while professional methods may create a satin finish.
- Professional nickel plating services can mix dyes into the coating, allowing for virtually any color.
Is it toxic to touch nickel?

No, but if you're allergic to nickel, you may experience a reaction. If you have a nickel allergy and touch a plated item, you could develop contact dermatitis. Your skin may break out in a rash, become itchy, or redden and dry out. These symptoms usually resolve on their own, but you should see a doctor if the condition worsens.
- Nickel is actually present in the air, water, and many household items, but in very small amounts. As long as you don't ingest or intentionally rub nickel into your skin, there is no cause for concern.
Is nickel plating better than chrome plating?
Nickel plating is ideal for protection, while chrome plating is primarily for aesthetic purposes. If the object will be exposed to chemicals or moisture, nickel plating is the better choice. While chrome is slightly harder than nickel, the difference in hardness is negligible, so you won’t notice much of a difference. The main reason to choose chrome plating is its appearance—it has a shinier, more reflective surface and can take on a bluish tint when light hits it.
- Chrome plating is usually more expensive, though the cost also depends on the nickel plating method you choose.
How can you remove nickel plating?

Purchase a non-toxic nickel stripper and immerse the object in it. Heat the nickel stripper according to the product instructions. Typically, you'll need to heat it to around 60°C to activate the solution. Then, dip the object into the solution for 10-15 minutes. Use tongs or a wooden spoon to remove the object and rinse it with clean water.
- You can buy nickel stripping solutions online.
- There are other methods for removing nickel plating, but they often require sulfuric acid and electrodes, making the process more complex and dangerous than using a nickel stripper.
