Rust is caused by the oxidation process of iron. The most common reason for rust formation is prolonged exposure to water. Any metal containing iron, including steel, reacts with oxygen atoms to form iron oxide, commonly known as rust. Rust accelerates corrosion, making regular maintenance crucial. Fortunately, removing rust isn't too difficult. This article will show you how to tackle the issue.
Steps
Solution Using Acidic Solution

Soak in Vinegar. Among the various home remedies, this non-toxic acid is highly effective at removing rust. Simply soak the rusty item in vinegar overnight and scrape it clean the following morning.
- Apple cider vinegar is recommended over white vinegar. White vinegar works too but is less effective than apple cider vinegar.
- Although vinegar is effective, its action is relatively mild. You might need to soak the item for more than one night; soaking for a whole day is ideal. Once you remove the rusty item from the vinegar, use crumpled aluminum foil soaked in vinegar to scrub off the rust.

Use lemon juice. Lemon juice is highly effective for removing rust from fabrics, but it can also work on metals when given enough time. Sprinkle salt on the rusted area, soak it in lemon juice, and then use crumpled aluminum foil to scrub the rust off.

Use a scientific method with phosphoric or hydrochloric acid. Phosphoric acid and hydrochloric acid are common, inexpensive household substances that are effective at removing rust. You can use them as follows:
- Phosphoric acid is a true "rust transformer", converting iron oxide (rust) into ferric phosphate, a black residue that forms on the surface. Soak the rusted object in phosphoric acid overnight, then allow it to dry. Once the surface is dry, scrape off the ferric phosphate. Phosphoric acid can be found in products like Coca-Cola, naval jelly, and rust removers.
- Hydrochloric acid is commonly used in the steel industry to "dip" steel to remove rust. It is found in many household cleaners, particularly toilet bowl cleaners.
- Hydrochloric acid continues to act even after the object is cleaned or dried. The acid's fumes can affect and discolor other metal or polished objects in the room. To prevent this, you can heat the treated object in an oven or on a stove. Another option is to neutralize the acid using a paste of lime or chalk.

Use potatoes. The oxalic acid in potatoes helps to remove rust. This method is particularly useful for smaller objects, such as knives. There are two ways to use potatoes to clean rust:
- Simply insert the knife into a potato and let it sit for a day or overnight. (Be cautious when inserting the knife into the potato). Afterward, remove the knife and scrape off the rust.
- Cut the potato in half, sprinkle a generous amount of baking soda inside, and vigorously scrub the rusted surface. Then, wipe clean using an abrasive material, such as a pot scrubber.

Check if you have other acids in your home. Sometimes, you can create your own rust-removing solution without leaving the kitchen. Any acidic substance will break down iron oxide and eventually clean the rust. Homemade solutions are especially helpful for small rusted items.
- The active ingredients in most store-bought rust removers are acids, usually phosphoric or hydrochloric acid, and household acidic substances can work similarly.
- If you're concerned about acid reactions or chemicals, quickly research online before using them. While most household substances can be safely combined, there are some interactions best avoided.

Clean rust with carbonated soda. Soak or dip the rusted object into a cup or bowl filled with carbonated soda. Check every 30 minutes. The soda may have an effect on the rust.
Solutions using paste mixtures

Create a paste with baking soda. Mix baking soda with water to form a paste that’s slightly thicker than toothpaste. The ratio of baking soda should be higher than the water. Apply this paste to the rusted object and scrub with an abrasive material like a pot scrubber or a toothbrush. Wipe clean and check.
- You may need to reapply the baking soda paste several times, but this method is definitely effective.

Make a mixture of hydrogen peroxide and cream of tartar. Create a paste with a consistency similar to the baking soda paste by using more cream of tartar than hydrogen peroxide. Apply it to the rusted surface, then scrub with an abrasive material and wipe clean.
- If hydrogen peroxide is unavailable, you can substitute it with water for similar results. The active ingredient that helps remove rust here is the cream of tartar.
Mechanical abrasion solutions
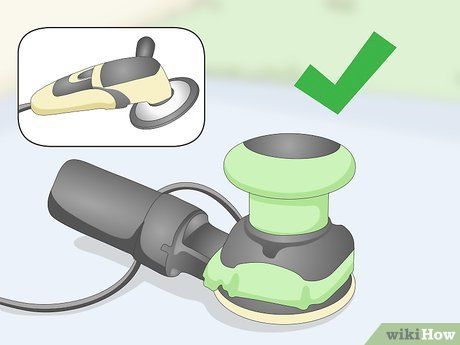
Purchase a grinder or polishing machine if you don't already have one. These machines are available at tool stores, though they can be quite expensive since they are power tools. However, many tool shops also offer these devices for rent at a relatively low cost. A grinder is particularly useful for larger surfaces, such as an old car's body.

Attach the coarsest grinding disc to the machine. The grinder is equipped with removable and replaceable discs that can be swapped out once they are worn and no longer effective. Coarse abrasive discs, fiber discs, and flap discs will work well.
- It's best to begin rust removal with the largest, coarsest disc to clean the heaviest rusted spots and avoid wearing down the smaller, finer discs.

Secure the rusted object during grinding. Clamp the object with a vice if possible, or ensure it is heavy enough to prevent movement while you work on it.

Switch on the grinder and let the disc gently sweep over the rusted area. Maintain steady movement to prevent damaging the underlying metal surface.

Use a polishing machine to finish off the rust cleaning process. If any small patches of rust remain, the polishing machine will eliminate them. The polishing machine functions similarly to the grinder, but it vibrates the abrasive pad rather than rotating a disc.
- Detail polishing machines are specially designed for polishing hard-to-reach areas and are ideal for removing rust from corners and rough surfaces.
Electrolysis solution

Create an electrolytic solution. At first, this method may sound challenging, but in reality, it's much easier than you think. Use a plastic bucket to hold the rusted object, fill it with enough water to submerge it, and then dissolve baking soda into the water at a ratio of 1 tablespoon of baking soda per 3.8 liters of water. Stir until fully dissolved.

Use a piece of steel as the sacrificial anode. The electrolysis process will separate the rust from the object you're cleaning and attach it to this metal piece. You need a piece of metal large enough to be submerged halfway in the water, with the other half connected to the positive terminal above the water surface. This is very important.
- The steel piece is suitable for use as the anode, as long as it's large enough to stick out of the water.
- Ensure the anode is made of magnetic metal to avoid confusing it with aluminum. You cannot use aluminum or stainless steel as the sacrificial anode during electrolysis.
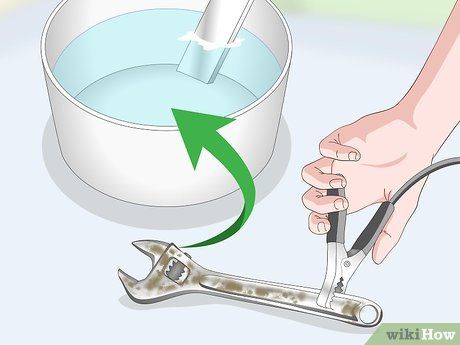
Securely attach the negative terminal (black) of the battery charger to the rust-free section of the object. You may need to scrape away the rust to expose a clean area. Submerge the object in water, making sure the cable remains above the water surface.
- Warning: Ensure the object does not touch the anode to prevent short-circuiting.

Connect the positive terminal (red) of the battery charger to the sacrificial metal piece. Be sure not to fully submerge the metal piece in water, as the positive terminal may corrode.
- If the sacrificial metal is completely submerged, you can use another metal wire to connect it to the car battery charger to keep the terminal and connection dry.
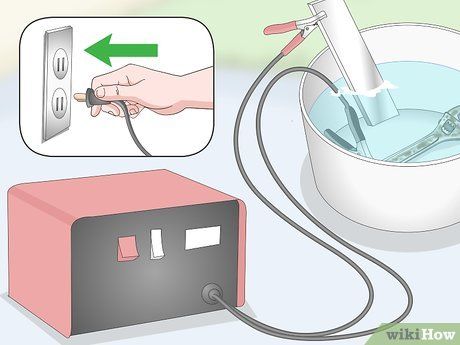
Plug in the car battery charger and switch it on. The electrolysis process will gradually clean the rust. Leave it for 12-20 hours.
- Warning: If you want to check the object, be sure to turn off and unplug the battery charger first. You will notice bubbles forming and dirt gathering on the surface. These are normal occurrences.
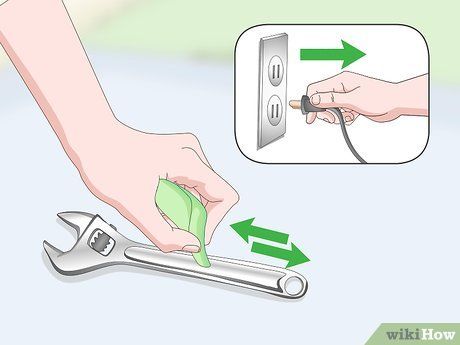
Disconnect the car battery charger and detach the cables from metal objects. Once removed, your item will no longer have rust, but you'll still need to clean it a bit further. Use a Scotch Brite abrasive pad to remove any leftover residue and a brush to clean those hard-to-reach spots.
Using commercial rust-removal chemicals

Purchase rust-removal chemicals. Indeed, there are rust-removal chemicals, but they often contain acids that may release toxic or semi-toxic gases. You can find these chemicals in hardware stores and some auto repair shops.
- Some available brands include Evapo-rust, Metal Rescue Rust Remover Bath (safe for paint, plastic, and leather), Acid Magic, The Works (20% HCL, hydrochloric acid), The Works Basic (9.5% HCL, hydrochloric acid), and WD-40 (light oil).
- Wear protective gear when working with rust-removal chemicals. This includes safety glasses, gloves, and a mask or face shield.

Apply the chemical solution. This is where the chemicals show their effectiveness. It will take time and effort to work their magic. You can choose from several methods:
- Some solutions come in spray bottles. Spray gently and precisely on the rusty areas and leave it overnight for heavily rusted spots.
- Other solutions need to be brushed on. Scrape off the easily removable rust and evenly apply the solution to the affected areas. Let it sit overnight.
- An alternative method is to soak the rusted item in the solution. For smaller items, you can use a plastic paint bucket or any suitable container, place the rusted object inside, pour the solution to cover it, and leave it overnight.
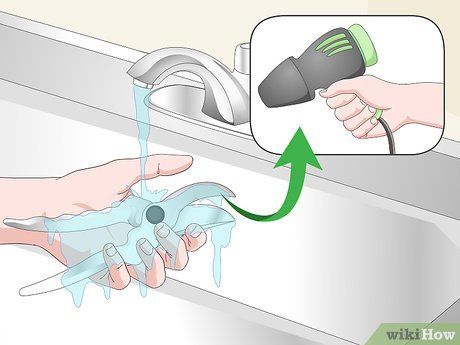
Rinse with water and let dry. Make sure to wash off the solution thoroughly. If possible, use a hair dryer to dry the object completely to prevent further rusting.

Scrape off any remaining rust. Rust that has settled overnight will begin to peel off, and the remaining parts can be easily scraped away.
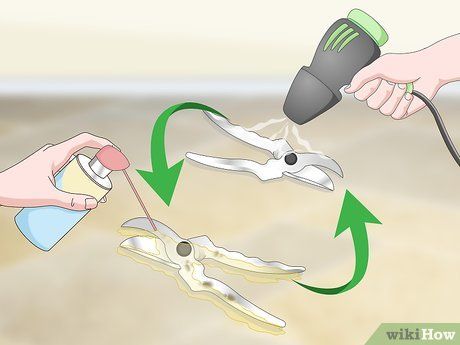
Repeat if necessary. The amount of time needed to clean rust depends on the object’s condition, the extent of rust, and the quality of the rust remover. Sometimes, you may need to apply the rust cleaner more than once, especially if the item is vertically positioned.
Advice
- Items that have been cleaned of rust may develop rust again. It's a good idea to protect your metal tools by applying a layer of oil. For other objects, consider applying a rust-resistant paint. If you're painting an item, make sure to apply at least one layer of rust-proof paint before the main coat.
Warning
- Irregular polishing and grinding actions may damage the metal underneath. If the item is valuable, consider using chemical solutions or electrolysis as an alternative method.
- Avoid inhaling strong acidic vapors; always ensure proper ventilation when working with acids. These fumes can irritate the throat and lungs, particularly for individuals with asthma or respiratory conditions. It’s advisable to wear protective gear such as goggles, gloves, and a mask. Always wear protective gloves when handling acidic products.
- Accurately identify the type of metal you are working with. Rust is another term for iron oxide, a substance that only forms on metals containing iron, such as steel. All metals are prone to corrosion in one way or another, and other metals have their own unique versions of rust. Some of the methods mentioned here, like electrolysis, are only effective on iron rust and should not be attempted for cleaning other types of metals.
- Exercise caution when using electrolysis for rust removal. You are dealing with electrical current. Ensure the container holding the water is made of an insulating material (plastic is the best), use rubber gloves, and avoid allowing the positive and negative wires to touch.
