Chemistry is an integral part of our lives, influencing everything from food preparation to vehicle operation, and even our bodily functions. We are immersed in an endless series of atomic rearrangements and energy exchanges, all driven by the principles of chemistry.
While the continuous flux of atoms often goes unnoticed in our day-to-day existence, certain chemical reactions have had a profound impact, altering the course of human history. Some have unlocked new possibilities, while others have revealed groundbreaking insights. Together, they mark significant milestones in humanity's development.
Below are 10 chemical reactions that have left a lasting mark on the world.
10. Ammonia Synthesis

Nitrogen is a crucial element for life, second only to carbon in importance. It forms an essential part of DNA, RNA, proteins, and chitin (a biological polymer found in fungi, insects, lobsters, shrimp, and some fish). Moreover, nitrogen is abundant on Earth, accounting for about 78% of the atmosphere. However, in its natural form, N2, nitrogen is inert and not usable by most life forms.
To become usable, nitrogen must be converted into more reactive forms like ammonia, nitrates, and nitrites. In nature, this transformation is typically carried out by specialized bacteria, which enter into a mutually beneficial relationship with plants, inhabiting nodules in their roots.
However, not all plants form this symbiotic relationship. In commercial agriculture, crops like corn do not fix nitrogen but instead absorb it from the soil. If such crops are cultivated for multiple seasons without nitrogen fixation, the soil will deplete its nitrogen reserves, making the use of fertilizer essential. Unfortunately, naturally occurring nitrogen sources are often insufficient to serve as effective fertilizers. This challenge necessitated the development of better methods for nitrogen fertilizer production to meet growing food demands.
The Haber-Bosch process marked a significant breakthrough. Invented by Fritz Haber and Carl Bosch in 1918, it used a combination of high temperatures, high pressures, and an iron catalyst to synthesize large quantities of ammonia from gaseous hydrogen and nitrogen.
Due to the relatively low cost of ammonia production, it quickly became a feasible substitute for natural fertilizers. Today, ammonia ranks as the second most produced chemical by tonnage, just behind sulfuric acid.
9. Polymerization of Polyethylene

Plastic has transformed the world. Its ability to be easily shaped, resistance to both heat and chemical damage, and affordability have made plastics an essential material in daily life—especially polyethylene. This versatile plastic is found in forms such as high-density polyethylene and low-density polyethylene, and is commonly used in products like plastic bags, milk bottles, and even bulletproof vests.
Polyethylene was discovered by chance in 1933 by two scientists at the Imperial Chemical Industries Research Laboratory. They were attempting to react ethylene and benzaldehyde, but instead stumbled upon a waxy material that was later identified as a polymer of ethylene. A polymer is a substance composed of repeating units, and other examples include cellulose and DNA.
By 1937, the material had been developed into a film and was used by the British during World War II for insulation in wires and radar components. Its lightweight nature allowed electrical components to be incorporated into planes, and its production methods were kept strictly confidential. Today, polyethylene holds the title of the most produced plastic globally, with 81.8 million tons produced in 2015 and nearly 100 million tons projected for 2018.
8. Combustion of Hydrogen

In the late 1700s, chemistry was still a relatively immature field. Most of its understanding was based on the Greek elements of air, water, earth, and fire, with new additions introduced to account for various observations.
One significant addition was the concept of phlogiston. Proposed by Georg Stahl, it suggested that all combustible substances contained a fire element called phlogiston, which was released into the air during combustion. This idea seemed to explain why burned charcoal weighed less than its original form. However, it couldn't explain why substances like phosphorus and sulfur increased in mass during combustion.
Enter Antoine Lavoisier, a French scientist who was highly doubtful of the phlogiston theory. In one of his most renowned experiments, he burned what was known as inflammable air (hydrogen gas) in the presence of normal air. The result was water. Lavoisier concluded that water must be a compound formed from a substance in the air, which he identified as oxygen, and the inflammable air.
Lavoisier further reinforced his theory by decomposing water into oxygen and hydrogen. In 1789, he published his groundbreaking work, *Traité élémentaire de chimie* ("Elements of Chemistry"), which rejected the ancient Greek system and established the principles that form the foundation of modern chemistry.
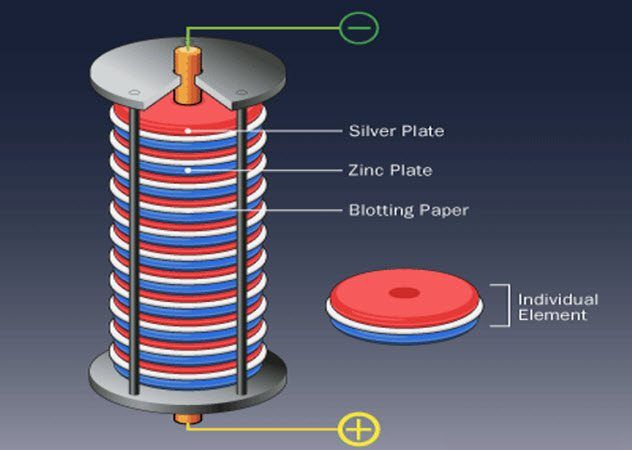
7. Reduction and Oxidation of Zinc and Silver
When Alessandro Volta was born in 1745 in Como, Italy, electricity was still a poorly understood phenomenon. It was known that electricity could be conducted and that it existed in two forms, which would later be recognized as positive and negative.
Shortly after Volta’s birth, Benjamin Franklin demonstrated that lightning was a form of electricity. Although Volta lacked a formal university education, he gained considerable recognition as a scientist. In 1775, he invented the perpetual electrophorus, an improvement upon earlier designs. However, his most important invention was still to come.
In 1780, Luigi Galvani, a scientist, claimed that animal muscles generated electricity when they contracted. He referred to this as "animal electricity" and believed it to be distinct from regular electricity.
Volta disagreed with this theory, pointing out that Galvani’s frog legs were connected to two different metals during the experiments. Volta then demonstrated that by stacking alternating silver and zinc metal discs, with brine-soaked cloths placed between each, he could generate a continuous electric current without the use of animals.
Volta’s invention quickly gained recognition as being far more valuable than just resolving his disagreement with Galvani. Prior sources of electricity could only produce short bursts. By generating a steady current, Volta's invention opened the door to more thorough studies, laying the groundwork for Faraday's pioneering work in electromagnetism.
6. Synthesis of Urea

Vitalism was the belief that living systems operated under completely different principles than nonliving ones. It was also thought that the components making up living systems could not be composed of nonliving materials. This idea was widely accepted in the 19th century and used to explain why many living systems appeared far more complex than nonliving ones.
German scientist Friedrich Wohler challenged this belief. Famous for isolating pure aluminum in 1825, Wohler was attempting to synthesize ammonium cyanate in 1828. However, when he reacted silver cyanate with ammonium chloride, he unexpectedly created white crystals. Upon further analysis, he identified the substance as urea.
Urea had been isolated in 1773 by French chemist Hilaire-Marin Rouelle, so Wohler had unintentionally synthesized an organic compound, disproving a core principle of vitalism. Wohler’s groundbreaking work ultimately laid the foundation for the development of organic chemistry.
5. PCR

Polymerase chain reaction (PCR) stands out as the most intricate reaction on this list, yet it holds the potential to be the most valuable and groundbreaking. Invented by Kary Mullis in 1983, PCR earned him a Nobel Prize for his pioneering work.
The process begins by heating DNA to separate it into two single strands. Since DNA is naturally double-stranded, primers can then attach to the individual strands. Enzymes known as DNA polymerases bind to the primers and replicate the remaining part of the DNA strand. This process can be repeated multiple times, with each cycle doubling the number of identical DNA copies.
The ability to replicate DNA has opened up vast possibilities across various fields. It has enabled forensic scientists to apply genetic techniques even with minute amounts of genetic material found at crime scenes. In medicine, it helps identify the origins of infections. In research, PCR was essential in sequencing the human genome.
Beyond these applications, PCR has become an essential technique in biology and biochemistry laboratories around the world.
4. Fat Hydration

Do you have a jar of Crisco in your kitchen? It might surprise you to learn that Crisco emerged from one of the most groundbreaking advancements in food technology.
The story begins with the distinction between animal fats and plant fats. Animal fats are typically saturated, meaning each carbon atom in the fat is bonded to the maximum number of other atoms. In contrast, plant fats are usually unsaturated, with some of the carbon atoms not fully bonded to the maximum number of atoms.
In 1902, Wilhelm Normann pioneered a method that allowed hydrogen to be added to unsaturated fats, converting them into saturated fats, or at least into more highly saturated fats. By 1909, Procter & Gamble purchased Normann's patent. Two years later, they launched Crisco, a shortening made primarily from hydrogenated cottonseed oil, which was more affordable than traditional lard.
But that was just the beginning. By 1979, nearly 60 percent of all fats consumed in the United States had been hydrogenated. However, hydrogenation had its drawbacks. Natural unsaturated fatty acids are almost always in the cis configuration, causing the fat molecules to bend or kink, making it harder for them to pack together. This is why most unsaturated fats are liquid.
However, during the hydrogenation process, some unsaturated fatty acids shift to the trans configuration. In the 1990s, studies revealed that consuming trans fats in high quantities led to negative health impacts. This prompted the FDA to regulate the amount of trans fats in foods, and certain regions even imposed bans on these substances. As a result, hydrogenated fats began to see a decline in usage.
3. Saponification

It's widely known that oil and water don't mix. This happens due to a property called polarity. Simply put, water molecules are polar, while oil molecules aren't. Because water molecules are polar, they prefer to stay close to each other rather than with nonpolar oil molecules. As anyone who's done dishes knows, this creates a challenge when cleaning – the grease refuses to mix with the water and remains on the dish.
The solution is soap. Soap molecules have both polar and nonpolar components. The polar section interacts with the water, while the nonpolar part bonds with the oil. This allows the oil to break into smaller droplets in the water, which are easier to wash away.
The chemical process used to make soap is known as saponification. In ancient times, soap was created by heating a mixture of salt, ashes, and animal fats in water. The earliest known soap-making occurred in Babylon around 2800 BC. Today, soap is made by reacting fatty acids (derived from fats) with either sodium hydroxide or potassium hydroxide.
However, soaps have been largely replaced by detergents for purposes beyond personal hygiene. While similar to soaps, detergents are typically derived from petrochemicals and offer several advantages. They are more durable and less likely to break down over time. Additionally, detergents are often more soluble in cold or hard water (which contains higher levels of calcium), meaning they are less likely to form the undesirable soap scum we often see.
2. Water with Carbon Dioxide

Carbon dioxide is most famous for its role as a greenhouse gas. As the concentration of carbon dioxide in the atmosphere increases, global temperatures also rise. But there’s another less-well-known side to carbon dioxide, and it plays a part every time we take a sip of soda.
When carbon dioxide dissolves in water, it forms carbonic acid, a reaction that can happen reversibly. Some of this carbonic acid then decomposes into bicarbonate and carbonate ions, releasing hydrogen ions (H+) in the process—an essential feature of Bronsted-Lowry acids. This acidic reaction contributes to the sharp taste we experience when drinking fresh soda.
Carbon dioxide can also interact with the water in the ocean in much the same way. In fact, the ocean absorbs around one-quarter of the carbon dioxide emitted each year.
As a result, the pH levels of surface ocean waters have dropped by about 0.1 pH units since the start of the industrial revolution, representing nearly a 30% increase in acidity. While this heightened acidity is advantageous to some organisms like algae and seagrasses, it poses a threat to others, including oysters, clams, shellfish, and coral.
One UN report projected that ocean acidification could result in financial losses of up to $1 trillion by the year 2100.
1. Ozone Destruction

Mechanical refrigeration technology had been in use since at least the 1870s. However, a major limitation was that many of the refrigerants used at the time were either extremely toxic or flammable. Unfortunately, it wasn't uncommon for people to lose their lives due to refrigerant leaks.
In response to this problem, Frigidaire, Dupont, and General Motors collaborated to create a much safer refrigerant. The result was Freon, a mixture of chlorofluorocarbons (CFCs). Freon was so safe that its inventor inhaled it directly and then blew it out onto a candle during a demonstration at the American Chemical Society.
However, CFCs had an unforeseen issue at the time. As refrigerators using CFCs became widespread, the chemical rapidly accumulated in the atmosphere. When exposed to ultraviolet light in the upper atmosphere, CFCs would often release a chlorine atom.
Chlorine is highly reactive and facilitates the breakdown of ozone (O3) into molecular oxygen (O2). Since catalysts do not get consumed in the reaction but instead accelerate the rate of it, a single CFC molecule could result in the destruction of thousands or even millions of ozone molecules, leading to a significant depletion of the ozone layer.
Today, CFCs are strictly controlled under the Montreal Protocol and are no longer used as refrigerants. They have been replaced by a similar group of compounds called hydrofluorocarbons (HFCs). Although HFCs also pose some challenges (such as being potent greenhouse gases), no new refrigerants have been developed that are both non-toxic and non-flammable.
