While chemicals are an essential part of nearly everything around and inside us, they often carry a negative reputation. Although some chemicals can be useful, every chemical has the potential to be dangerous under certain conditions. However, the chemicals listed here are hazardous even when conditions seem ideal.
10. Ethidium Bromide
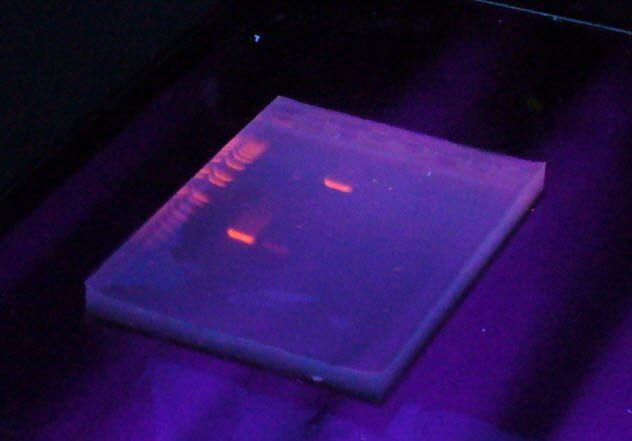
To be a biologist in today's world, mastering the techniques of working with DNA is essential. The challenge, however, lies in the fact that DNA is not visible at the concentrations commonly used in most experiments. If you need to isolate DNA fragments, dyeing it is a necessary step.
Ethidium bromide is a perfect dye for DNA. It fluoresces brightly and binds firmly to DNA. What more could you ask for? Perhaps a compound that doesn't cause cancer?
Ethidium bromide stains DNA by inserting itself between the base pairs. This action causes breaks in the DNA structure, as the presence of ethidium bromide induces strain. These breaks can serve as sites for mutation.
Mutations are generally unwanted. Additionally, to visualize the dye, you need to use UV light, another cancer-causing agent, which makes ethidium bromide no less dangerous. As a result, many researchers today prefer using safer compounds for DNA staining.
9. Dimethylcadmium
Lead, mercury, and their elemental counterparts pose various health risks when they enter the human body. In certain forms, heavy metals can pass through the body without being absorbed, while in others, they are easily taken up. Once inside, serious issues begin to arise.
Dimethylcadmium can cause severe burns to the skin and damage to the eyes. It is also an environmental toxin that accumulates in tissues. As if its physiological effects weren’t harmful enough, the chemical is highly flammable in both its liquid and gaseous forms. Exposure to air is enough to ignite it, and when it contacts water, it produces even more flammable compounds.
Burning dimethylcadmium produces cadmium oxide—another dangerous substance. In addition to being a carcinogen, exposure to cadmium oxide can lead to a flu-like condition known as “metal fume fever.”
8. VX
VX, also known as Venomous Agent X, is a chemical with no purpose other than chemical weaponry. It was developed by the British at the Porton Down research facility, and it is an odorless, tasteless substance that is lethal in quantities as small as 10 milligrams. The British government exchanged information about VX with the US in return for knowledge on thermonuclear weaponry.
VX is easily absorbed through the skin. It doesn’t degrade quickly in the environment, meaning an attack involving VX could lead to prolonged exposure risks. Clothing worn during exposure could be enough to cause toxicity in anyone who comes into contact with it. VX kills by continuously overstimulating glands and muscles. Death occurs when the respiratory system fails.
7. Sulfur Trioxide

Sulfur trioxide is a key precursor to sulfuric acid and is essential for certain sulfonation reactions. If it weren’t useful, no rational scientist would keep it on hand. As demonstrated in the video above, sulfur trioxide is extremely corrosive when it comes into contact with organic matter.
When sulfur trioxide interacts with water (which is abundant in the human body), it produces sulfuric acid and heat. Even if sulfur trioxide doesn’t directly touch your skin, simply being close to the reaction is hazardous. The fumes from sulfuric acid are harmful to the lungs. Spilling sulfur trioxide on organic materials like paper or wood can ignite a toxic fire.
6. Batrachotoxin

Batrachotoxin is a complex molecule so potent that just 136 millionths of a gram can be fatal to a 68-kilogram (150 lb) human. To put this into perspective, that is about the weight of two grains of salt. This makes batrachotoxin one of the most deadly chemicals known.
Batrachotoxin attaches to the sodium channels in nerve cells, which play a crucial role in muscle and nerve activity. By forcing these channels to stay open, the toxin disables all muscle control in the organism.
Batrachotoxin is found on the skin of small poison arrow frogs. Some Native American tribes have historically used this toxin by dipping their darts in the poison secreted by these frogs. A single dart hit incapacitates the prey, allowing hunters to approach and capture it at their convenience.
5. Dioxygen Difluoride
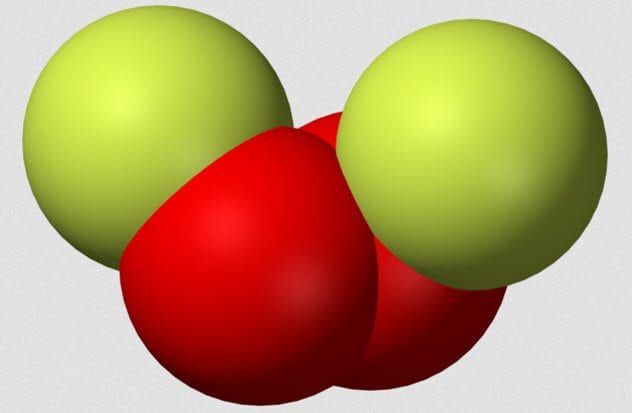
Dioxygen difluoride, also known by the intriguing nickname FOOF, is a highly dangerous chemical composed of two fluorine atoms connected by two oxygen atoms. In 1962, chemist A.G. Streng published a paper titled “The Chemical Properties of Dioxygen Difluoride.” While the title may seem dry, Streng's experiments were far from boring.
FOOF must be synthesized at extremely low temperatures, as it breaks down when heated to its boiling point of about -57 degrees Celsius (-71 °F). During his research, Streng discovered that FOOF reacted violently with organic compounds, even at -183 degrees Celsius (-297 °F). When combined with chlorine, FOOF triggered a catastrophic explosion, and a similar reaction occurred with platinum.
In fact, the results section of Streng's paper is filled with words like “flash,” “spark,” “explosion,” “violent,” and “flame,” all in various combinations. Keep in mind, these reactions were taking place at temperatures where most chemicals remain stable and inert.
4. Potassium Cyanide
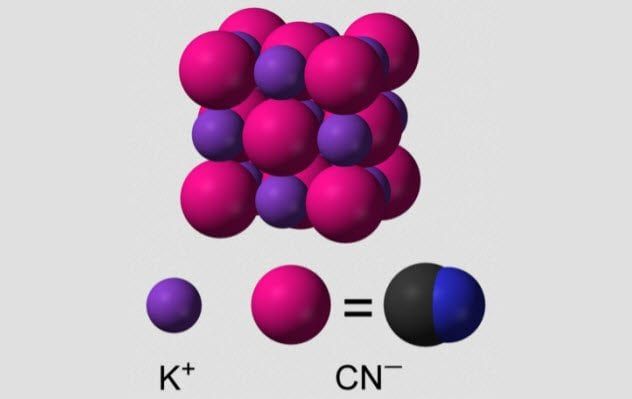
Cyanide is a straightforward molecule—just a carbon atom triple-bonded to a nitrogen atom. Due to its small size, cyanide can infiltrate many parts of proteins, and unfortunately, it never works out well for us. Cyanide is especially drawn to bond with the iron atoms found in hemeproteins.
One important hemeprotein is hemoglobin, the protein responsible for transporting oxygen in our blood. Cyanide disables hemoglobin’s ability to carry oxygen, effectively hindering this vital function.
When potassium cyanide encounters water, it decomposes into hydrogen cyanide, a gas that the body easily absorbs. This gas has the distinct smell of bitter almonds, though not everyone can detect it.
Because of its rapid effects, potassium cyanide has been the preferred chemical for suicide by many. During World War II, British agents carried potassium cyanide pills as a precaution against capture, and several high-ranking Nazis resorted to it as a means of evading justice.
3. Hydrofluoric Acid

Hydrofluoric acid is a substance that everyone working in chemistry has heard ominous tales about. Technically, hydrofluoric acid is a weak acid that doesn't readily release its hydrogen ion, meaning it doesn’t cause an immediate chemical burn.
This characteristic makes hydrofluoric acid particularly dangerous. It is relatively neutral, allowing it to pass through the skin unnoticed and enter the body. Once inside, hydrofluoric acid begins its damaging work.
Once the acid releases its proton, the resulting fluorine reacts with other substances. These reactions are typically brief, with the fluorine quickly causing harm before being released to continue its destructive work.
Fluorine has a particular affinity for calcium. As a result, hydrofluoric acid induces bone destruction. It also depletes the calcium needed for proper heart function. Without treatment, the outcome is often a slow and painful death.
2. Chlorine Trifluoride
Individually, chlorine and fluorine are already hazardous elements. However, when combined to form chlorine trifluoride, they become even more dangerous.
Chlorine trifluoride is so corrosive that it cannot be contained in glass. It is an extremely potent oxidizer, capable of igniting materials that would not catch fire in pure oxygen.
Even the remains of objects that have already burned in oxygen will ignite upon contact with chlorine trifluoride. No external ignition source is required. In an industrial accident where 900 kilograms (2,000 lbs) of chlorine trifluoride spilled, it corroded 0.3 meters (1 ft) of concrete and 1 meter (3 ft) of gravel beneath it.
The safest (relatively) way to store this chemical is in a metal container that has already been fluorine-treated, forming a fluoride barrier that prevents chlorine trifluoride from reacting with the metal.
When chlorine trifluoride comes into contact with water, it reacts violently. The chemical produces intense heat and hydrofluoric acid.
1. Dimethylmercury

Just two drops of dimethylmercury can be deadly—and they have been.
In 1996, Karen Wetterhahn was researching the impact of heavy metals on living organisms. Although metals in their solid form are difficult for organisms to absorb, it is still possible to dip your hand in liquid mercury with minimal harm—though this is certainly not advised.
To introduce mercury into DNA, Wetterhahn used dimethylmercury, a compound with a mercury atom bonded to two organic groups. During her research, she accidentally spilled one, possibly two, drops onto her latex glove. Within half a year, she was dead.
Despite being an experienced professor who followed all safety guidelines, the dimethylmercury passed through her gloves in less than five seconds, penetrating her skin in just 15. The chemical left no visible traces, and she only began to notice symptoms months later, by which time it was too late for treatment.
A colleague who witnessed Wetterhahn in her final moments recalled: 'She was thrashing about. Her husband saw tears rolling down her face. I asked if she was in pain. The doctors said it didn’t appear that her brain could even register pain.'
