
How familiar are you with the periodic table? Our series The Elements delves into the core building blocks of the universe—and how they connect to your daily life—one element at a time.
Silicon is a metalloid, a unique element that doesn't fully resemble metals or non-metals. Whether it’s the smartphone in your pocket or the dust on your shoes, silicon is part of your world. Discover more about this ever-present element.
1. IT'S EVERYWHERE YOU LOOK.
Silicon is the seventh most abundant element in the cosmos and even more abundant in Earth's crust, coming in second to oxygen as the most common element by weight. The mantle beneath the crust is also rich in silicon. With an atomic number of 14, it lies just below carbon in the periodic table.
2. SILICON AND SILICONE ARE NOT THE SAME.
 iStockphoto
iStockphotoYou might associate the term silicone with breast implants, but it actually refers to a group of synthetic materials made from alternating silicon and oxygen atoms, with carbon and hydrogen molecules bonded to the sides. By modifying these side groups and creating links between chains, chemists can craft silicones with various properties. While silicones are used in breast implants, they also appear in car polish, electric cable insulation, and even your hair conditioner, where they help tame frizz. And let’s not forget Silly Putty, invented during World War II when scientists were trying to develop an alternative to rubber—and instead created a new favorite toy.
3. HUMANS HAVE USED SILICON FOR MILLENNIA WITHOUT KNOWING ITS TRUE NATURE.
Silica is the key component in glass, a material that humans have been crafting for thousands of years, dating back to the Egyptians, who created beads from it around 2500 BCE. In China, during the Qin and Han dynasties, barium copper silicates were used to create purple and blue pigments for various decorative purposes, including parts of the iconic terra-cotta army.
It took centuries for people to realize that silica could be split into two distinct elements—oxygen and silicon. In the late 1700s, French chemist Antoine Lavoisier noticed that certain substances classified as 'earth' materials (dry and cold) sometimes displayed metal-like characteristics (such as hardness, density, and resistance to stretching). Silica was one of these. Lavoisier theorized that some of these 'earths' might actually be molecules of oxygen combined with an undiscovered, metal-like element.
At the time, chemists didn't yet know how to separate the oxygen atoms, which form strong bonds with silicon. That changed in the 1820s when Swedish chemist Jons Berzelius succeeded in isolating silicon in his lab by purifying it from a silicon-containing compound. (The exact compound and method remain unclear.) Unfortunately, Berzelius's discovery came too late for Lavoisier, who had passed away in 1794 and missed seeing his ideas confirmed.
4. SILICA IS THE MOST COMMON FORM OF SILICON.
Known also as silicon dioxide, this molecule consists of one silicon atom and two oxygen atoms (SiO2). Most of the silicon we encounter is actually silica, which can be found in both minerals and plants. Many plants form tiny structures called phytoliths from the silica they absorb from the soil. The reason for this is still unclear—these phytoliths might provide protection from microscopic threats, offer structural support, or simply serve as a way for plants to dispose of excess silica.
Phytoliths can persist long after plants decay, offering insights into the ancient history of a region—such as whether it was once a forest or grassland, or how humans interacted with the environment. Dan Cabanes, a phytolith specialist and anthropologist at Rutgers University, has utilized these microscopic plant remains to uncover how Neanderthals established their living spaces in a cave in northern Spain, crafting a bedding area with grass that was used repeatedly. Moreover, because phytoliths endure even after being exposed to fire, Cabanes explains that they can help us understand the types of food Neanderthals ate and the fire-making techniques they employed.
The analysis isn't always flawless, however, since sometimes different plant species produce phytoliths that look alike, and there are even some plants that don't create them at all.
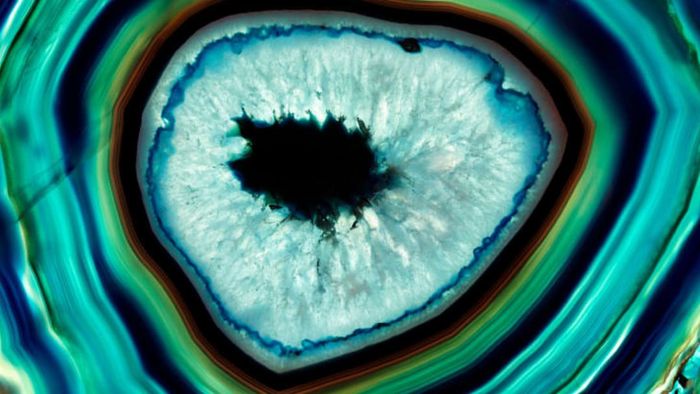
5. A CRUCIAL ELEMENT IN SOME STUNNING GEMSTONES …
 iStock
iStockBeautiful gemstones such as amethyst, onyx, and agate are primarily composed of silica. Within each of these stones, the silica molecules form intricate, repeating 3D patterns known as crystal structures. Variations in these structures, as well as the presence of minor impurities, contribute to the unique visual qualities of each gemstone.
6. … AND THE STUNNING ELEGANCE OF DIATOMS.
 Anatoly Mikhaltsov, Wikimedia Commons // CC BY-SA 4.0
Anatoly Mikhaltsov, Wikimedia Commons // CC BY-SA 4.0Silica also forms the cell walls of diatoms, a fascinating form of algae found across the globe. These diatoms come in an astonishing range of shapes and can thrive in both fresh and saltwater. After they die, their cell walls gather together to form chalky layers of 'diatomaceous earth,' a substance used in a variety of products, from cat litter to toothpaste.
7. SILICON IS ESSENTIAL IN MODERN TECHNOLOGY …
Silicon functions as a semiconductor—a material that doesn't fully conduct electricity nor completely insulate it, but instead lies in between. This feature is crucial for various electronic devices, where controlling the flow of electricity is necessary. Eric Pop, a professor of electrical engineering at Stanford University, explains to Mytour, “What’s remarkable about semiconductors is their conductivity can be adjusted by introducing small amounts of other elements.” Pure silicon is an insulator, but by ‘doping’ it with minute quantities of elements like phosphorus or arsenic, it becomes much more efficient at conducting electricity.
Other substances, such as germanium or gallium arsenide, are more effective semiconductors than silicon, but silicon remains the most widely used material among electronics producers (whose concentration south of San Francisco during the 1970s led to the creation of the term 'Silicon Valley'). It's affordable, abundantly available, and because of its strong tendency to oxidize, it conveniently forms its own insulating layer when exposed to air.
8. … BUT SCIENTISTS ARE ON THE LOOKOUT FOR IMPROVED ALTERNATIVES.
Engineers like Pop are exploring other materials that could replace silicon in electronic devices, aiming to meet the ever-growing demand for faster processing speeds. As Pop puts it, “Silicon is kind of like the Honda Civic of semiconductors. It gets the job done, but it’s not the fastest.” Nevertheless, Pop believes that even with the emergence of superior materials, silicon will not be entirely phased out, due to its low cost.
9. SILICON SUPPORTS MANY OF OUR STRUCTURES.
 iStock
iStockMany everyday construction materials are derived from substances that contain silicon. For instance, clay minerals, which are silicon-rich, are used in the production of bricks and Portland cement, which then serves as the binding agent in concrete.
10. AMERICANS LEFT A PIECE OF SILICON ON THE MOON.
When Buzz Aldrin and Neil Armstrong became the first humans to set foot on the Moon in 1969, they left more than just footprints behind. Among the items they left was a small silicon disc, engraved with messages from the leaders of 73 nations, ranging from Afghanistan to Zambia. The disc, protected by an aluminum case, was stored in a small bag along with other items. Silicon was chosen for this role due to its ability to withstand the extreme temperature variations on the Moon. The disc almost didn’t make it, though, as Aldrin had forgotten about the bag, which was tucked into a pocket of his spacesuit sleeve. He was already on the ladder to the spacecraft when Armstrong reminded him, prompting Aldrin to toss the pouch onto the lunar surface.
