While most people can quickly recall the three traditional states of matter—solid, liquid, and gas—those with a bit more scientific knowledge often recognize plasma as a fourth. However, scientists have broadened the list of potential states of matter far beyond these four. Through their discoveries, we've gained insights into the origins of the universe, lightsabers, and even a mysterious state of matter found in something as simple as a chicken.
10. Non-Crystalline Solids

Non-crystalline solids are a captivating category of the solid state. Unlike typical solids, where the molecules are highly ordered and unable to move freely, non-crystalline solids exhibit a structure somewhere between solids and liquids. When a liquid undergoes a process called vitrification, it cools and its viscosity increases, preventing it from flowing like a liquid. However, the molecular structure remains disordered and doesn't form the regular crystalline structure of typical solids.
The most well-known example of an amorphous solid is glass. For centuries, glassmakers have crafted glass using silica. When they cool the silica from its liquid state, it doesn’t solidify immediately after reaching its melting point. As the temperature continues to drop, the viscosity increases, giving the glass the appearance of being solid. However, the molecular structure remains disordered. This process turns glass into an amorphous solid. This transformation has enabled artists to create stunning and surreal glass sculptures.
So, what’s the real difference between an amorphous solid and a regular solid? In daily life, not much. Glass appears completely solid unless you look closely at its molecular makeup. And don’t believe the myth that glass flows like liquid over time. Some tour guides claim that old glass in churches appears thicker at the bottom due to its flow, but this is actually due to imperfections in the glassmaking process, with the thicker side being naturally placed at the bottom. While this myth might be boring, studying materials like glass has led researchers to new understandings of phase transitions and molecular structures.
9. Supercritical Fluids

Most phase transitions happen under specific temperature and pressure conditions. We all know that raising the temperature will eventually turn a liquid into a gas. However, when both pressure and temperature increase, the liquid enters the realm of supercritical fluids, which possess characteristics of both gases and liquids.
For example, supercritical fluids can pass through solids like a gas but also function as a solvent like a liquid. Remarkably, the properties of a supercritical fluid can be adjusted to be more gas-like or more liquid-like depending on the balance of pressure and temperature. This ability has allowed scientists to develop a range of applications for supercritical fluids, from the highly specialized to the everyday.
While supercritical fluids may not be as familiar as amorphous solids, you likely come across them just as often as you do glass. Supercritical carbon dioxide has become popular among brewing companies for its effectiveness as a solvent in hop extraction, and coffee producers use it to create high-quality decaffeinated coffee. Supercritical fluids are also utilized to improve hydrolysis efficiency and enable power plants to operate at higher temperatures. Despite being an unfamiliar state of matter, you probably encounter products derived from supercritical fluids every day.
8. Degenerate Matter

While amorphous solids are found on Earth, degenerate matter exists only in certain types of stars. It arises when the outward pressure of matter is not controlled by temperature, as it is on Earth, but by intricate quantum laws, particularly the Pauli exclusion principle (we’ll explore this shortly). This means that the outward pressure of degenerate matter remains even if its temperature drops to absolute zero. The two primary forms of degenerate matter are electron-degenerate matter and neutron-degenerate matter.
Electron-degenerate matter is mainly found in white dwarf stars. It forms in the star's core, where the surrounding matter's weight compresses the core's electrons into their lowest energy state. However, according to the Pauli exclusion principle, no two electrons can occupy the same energy state. As a result, the electrons push against the surrounding material, creating outward pressure due to the quantum laws that prevent all electrons from occupying the lowest energy state. This phenomenon can only persist if the star's mass is less than 1.44 times that of the Sun, known as the Chandrasekhar limit. If a star exceeds this mass, it will collapse into a neutron star or a black hole.
When a star collapses into a neutron star, it transforms from having electron-degenerate matter to consisting of neutron-degenerate matter. Due to the immense gravity of a neutron star, the electrons combine with protons in the core, resulting in neutrons. Free neutrons, which are not bound within an atomic nucleus, typically have a half-life of 10.3 minutes. However, in the core of a neutron star, the star's mass enables neutrons to exist outside of a nucleus, creating neutron-degenerate matter.
There may be other exotic forms of degenerate matter, such as strange matter, which could exist in a rare type of star known as a quark star. Quark stars are a transitional phase between neutron stars and black holes, where the quarks in the core separate and form a mixture of free quarks. These stars have not yet been observed, but physicists continue to theorize their potential existence.
7. Superfluid

Let’s return to Earth to explore superfluids. A superfluid is a phase of matter that occurs when certain isotopes of helium, rubidium, and lithium are cooled to nearly absolute zero. While it shares similarities with a Bose-Einstein condensate (BEC), there are subtle distinctions. Some Bose-Einstein condensates are superfluids, and some superfluids are BECs, but not all from either category fit the other.
The most commonly observed superfluid is liquid helium. When helium is cooled to the 'lambda point' of 2.17 degrees Kelvin, part of the liquid transitions into a superfluid. Most substances, when cooled to a certain point, will form a solid as atomic attractions overcome thermal vibrations. However, helium atoms interact so weakly with one another that it remains a liquid all the way down to absolute zero. At this temperature, the characteristics of the atoms merge, leading to the unusual properties of superfluids.
To begin with, a superfluid lacks internal viscosity. When placed in a test tube, superfluids will climb the walls of the tube, appearing to defy gravity and surface tension. Liquid helium is notorious for leaking easily, as it can seep through even the smallest cracks. Superfluids also display unusual thermodynamic characteristics. They possess zero thermodynamic entropy and can conduct heat infinitely. As a result, if heat is introduced, it spreads so rapidly that it generates thermal waves, a phenomenon not observed in normal liquids.
6. Bose-Einstein Condensate

Bose-Einstein condensates are among the most well-known but also the most difficult-to-grasp states of matter. To understand them, we first need to distinguish between fermions and bosons. Fermions are particles with half-integer spins (like electrons) or composite particles (like protons). These particles follow the Pauli exclusion principle, which governs electron-degenerate matter. In contrast, bosons have integer spins, and multiple bosons can occupy the same quantum state. Bosons include force-carrying particles, such as photons, and certain atoms, like helium-4, known as bosonic atoms.
In the 1920s, Albert Einstein used the research of Indian physicist Satyendra Nath Bose to propose a new type of matter. Einstein's theory suggested that cooling certain gases to just above absolute zero would cause their wave functions to merge into a single 'superatom,' exhibiting quantum effects on a macroscopic scale. However, it wasn't until the 1990s that technology advanced enough to cool elements to the required temperatures. In 1995, researchers Eric Cornell and Carl Wieman managed to coalesce 2,000 atoms into a Bose-Einstein condensate, large enough to be viewed under a microscope.
Bose-Einstein condensates are closely related to superfluids, though they have distinct properties. One of the most remarkable is their ability to slow light from its usual speed of 300,000 kilometers per second. In 1998, Harvard researcher Lene Hau managed to reduce the speed of light to a mere 60 kilometers per hour (37 mph) by passing a laser through a cigar-shaped sample of BEC. Later experiments by Hau’s team even succeeded in halting light completely by switching off the laser as it passed through the BEC. These breakthroughs have paved the way for advancements in light-based communication and quantum computing.
5. Jahn-Teller Metals

Jahn-Teller Metals are a newly discovered state of matter, with their first successful creation only occurring in 2015. If subsequent studies confirm the findings, this breakthrough could revolutionize our understanding of materials, as Jahn-Teller metals possess characteristics of both insulators and superconductors.
Led by chemist Kosmas Prassides, researchers experimented by inserting rubidium into carbon-60 molecules (commonly known as buckyballs), forcing the molecules to adopt a new shape. The resulting metal is named after the Jahn-Teller effect, which explains how pressure can alter the molecular geometry, leading to new electron configurations. In chemistry, pressure is not only the result of compression but can also arise from the addition of new atoms or molecules, fundamentally changing the material's properties.
When Prassides's team introduced rubidium into the carbon-60 molecules, the material shifted from being an insulator to a superconductor. However, due to the Jahn-Teller effect, the molecules resisted this change and clung to their original configuration, resulting in a substance that appears insulating yet possesses the electrical properties of a superconductor. This transition from an insulator to a superconductor had never been observed before these experiments.
What makes Jahn-Teller metals particularly exciting is their ability to become superconductors at much higher temperatures (–135 degrees Celsius, compared to –243.2 degrees Celsius). This brings them closer to achievable conditions for mass production and practical experimentation. If these findings hold true, we could soon be on the path to mass-producing materials that conduct electricity without resistance—without generating heat, sound, or energy loss—potentially revolutionizing energy production and transportation.
4. Photonic Matter

For years, it was believed that photons were massless and did not interact with one another. However, recent breakthroughs by researchers at MIT and Harvard have demonstrated that light can exhibit mass-like properties—and they've even created 'light molecules' that can bounce off each other and bond. While this may sound underwhelming, it's actually the first step toward creating a lightsaber.
The science behind photonic matter may seem complex, but bear with it. (Remember, lightsabers!) The process began with experiments involving supercooled rubidium gas. When a photon is passed through this gas, it interacts with the rubidium molecules, losing energy and slowing down in the process. Ultimately, the photon emerges from the cloud, significantly slower but still maintaining its identity.
Things take a strange turn when two photons are directed through the gas, triggering what’s called the Rydberg blockade. When one atom is excited by a photon, nearby atoms cannot be excited in the same way. In essence, the first excited atom blocks the path of the photons. For a surrounding atom to be excited by the second photon, the first photon must continue its journey through the gas. Normally, photons don’t interact, but under the influence of the Rydberg blockade, they push one another through the gas, exchanging energy and interacting along the way. From an external viewpoint, these photons appear to gain mass and behave as a single entity, even though they remain massless. When they exit the gas, they appear to be joined together, almost like a molecule of light, capable of being deflected and shaped.
Practical uses for photonic matter may be far off, but researcher Mikhail Lukin already has a long list of potential applications. These range from computing and creating three-dimensional crystals made entirely from light, to—yes—crafting lightsabers.
3. Quark-Gluon Plasma
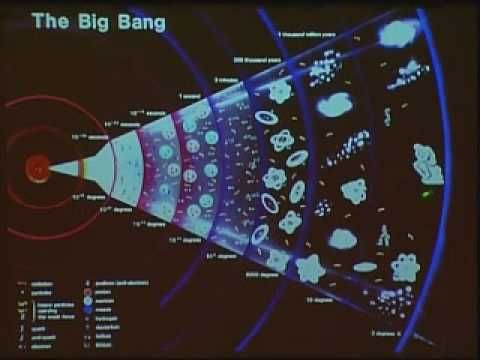
For our final look at obscure states of matter, let’s revisit the one that we all originated from: quark-gluon plasma. In fact, the early universe existed in a completely different state of matter than what we consider normal today. But first, a little context.
Quarks are the fundamental particles that make up hadrons, such as protons and neutrons. Hadrons are composed either of three quarks or one quark and one anti-quark. Quarks carry fractional charges and are bound together by gluons, the exchange particles for the strong nuclear force.
Free quarks do not appear in nature, but right after the Big Bang, they existed briefly for about a millisecond alongside gluons. During this incredibly hot period, the quarks and gluons interacted very little, moving at near-light speed. The entire universe was made up of this hot quark-gluon plasma. A fraction of a second later, as the universe cooled, it allowed for the formation of heavy particles like hadrons, and quarks began interacting with each other and gluons. This moment marked the beginning of the universe as we know it, with hadrons combining with electrons to form the first primitive atoms.
In today’s universe, scientists have attempted to recreate quark-gluon plasma in large particle accelerators. In these experiments, hadrons collide with each other, generating the high temperatures required for quarks to temporarily decouple. These experiments have provided us with insights into the properties of quark-gluon plasma, revealing that it was frictionless and resembled a liquid more than the plasmas we are familiar with. As research continues, we will gain more understanding of how and why the universe took the shape it did.
2. String-Net Liquid

Have you ever wondered what the vacuum of space is made of? It’s a question many don’t often consider, but in the last decade, MIT’s Xiao-Gang Wen and Harvard’s Michael Levin proposed a novel state of matter that could help us uncover fundamental particles beyond the electron.
The development of the string-net liquid model traces back to the mid-1990s when a group of scientists proposed the existence of "quasi-particles" after observing an experiment in which electrons passed between two semiconductors. This discovery caused a sensation, as these quasi-particles appeared to possess fractional charges—something physics at the time deemed impossible. The team theorized that the electron might not be a fundamental particle and suggested there were more basic particles yet to be discovered. Their work earned them a Nobel Prize, but it was later revealed that their findings were based on an experimental error. As a result, the concept of "quasi-particles" was abandoned.
Some researchers, however, were not ready to abandon the idea. Wen and Levin took the concept of quasi-particles and proposed a new state of matter known as the string-net. In this model, quantum entanglement serves as the fundamental property. While it might appear chaotic at a microscopic level, the string-net exhibits a high level of order when considered as a whole, thanks to the entanglement of its electrons. Wen and Levin further extended their research to include other particles and their entanglement behaviors.
In their computer simulations of the new state of matter, Wen and Levin discovered that the ends of a string-net could generate various subatomic particles, including the elusive "quasi-particle." Even more astonishing, they found that when string-nets vibrated, they did so in accordance with Maxwell's equations, the principles that govern light. In their papers, they proposed that the vacuum of space is filled with string-nets of entangled subatomic particles, with the ends of these strings representing the subatomic particles we observe. They also suggested that this string-net liquid might be the very source of light, potentially offering a way to unify matter and light.
While this theory might seem far-fetched, it is worth noting that in 1972, long before the string-net concept, geologists discovered an unusual mineral in Chile called herbertsmithite. In this mineral, electrons form triangular structures, which contradicts our understanding of electron interactions. Remarkably, this triangular arrangement is predicted by the string-net model, and researchers have worked with synthetic herbertsmithite in an effort to verify the model's accuracy. However, the question of whether this theoretical state of matter truly exists remains unresolved.
1. Disordered Hyperuniformity

In 2003, Salvatore Torquato and Frank H. Stillinger of Princeton University introduced a groundbreaking state of matter called disordered hyperuniformity. Though this may sound contradictory, the concept is that the material appears disordered when viewed up close but exhibits hyperuniformity and long-range order. This type of matter combines characteristics of both crystals and liquids. Initially, it was thought to exist only in simple plasmas and liquid hydrogen. However, researchers recently made an unexpected discovery of this state in the eye of a chicken.
Chickens have five types of cones in their eyes, four of which detect color and one that detects light levels. Unlike the human eye or the hexagonal eyes of insects, the cones in a chicken’s eye appear randomly dispersed without any clear order. This randomness is due to an exclusion zone around each cone that prevents adjacent cones of the same type. As a result, the cones cannot form a crystalline structure typical of solids. However, when the cones are viewed collectively, they form a highly ordered pattern, as shown in Princeton’s images. Thus, the cones in a chicken’s eye behave like a liquid when observed up close and like a solid from a distance. This is distinct from amorphous solids, where the material remains unordered at all scales.
Ongoing research is exploring this new state of matter, which may be more widespread than originally believed. Currently, Princeton researchers are investigating the potential applications of hyperuniform materials in creating self-arranging structures and light detectors designed for specific wavelengths.
