The world is far from perfect, and tragedies that could have been avoided are unfortunately inevitable. However, some incidents arise from obvious mistakes that could have been prevented with even a bit more attention. In healthcare, multiple precautions are in place to avoid contamination and infection, but there have been situations where these safeguards failed.
10. Antibiotic-Resistant Duodenoscopes

Duodenoscopes are medical tools designed to drain fluids from the pancreatic and bile ducts, but they are notoriously difficult to sterilize. These devices feature an elevator-like mechanism that enables them to navigate within the body, draining fluids as necessary. This complexity makes proper cleaning quite challenging. While most skilled medical professionals would clean such instruments thoroughly, many failed to do so, which led to fatal consequences.
In 2016, two people in the Chicago area succumbed to a bacterial infection that had been spreading across the United States. From 2012 to 2015, approximately 250 individuals contracted this illness, which was traced back to a defect in the same device: the duodenoscope.
The manufacturers of the duodenoscope never actually tested its cleaning procedures in real-world conditions. As a result, bacteria spread that otherwise wouldn’t have proliferated if the devices had been properly sterilized. The infectious agent responsible was a superbug known as carbapenem-resistant Enterobacteriaceae (CRE).
CRE is an extremely lethal bacterial strain that kills half of those it infects and is resistant to even the most powerful antibiotics. CRE and similar drug-resistant outbreaks of E. coli have been reported in the US and Europe, notably in France and Germany. Due to the manufacturers' failure to disclose the device's flaws, Olympus, Pentax, and Fujifilm were investigated by the FDA, and hospitals were cautioned against using these scopes.
9. Drug-Contaminated Diet Supplements

In 2013, the FDA tested a dietary supplement called Dr. Larry’s Tranquility pills, which claimed to induce natural sleep using ingredients like figwort root and licorice. However, the pills were found to contain much more than expected, with potent prescription drugs included.
Two powerful sedatives were identified: Thorazine, an intense antipsychotic, and doxepin, an antidepressant and sleep aid. It was also discovered that Dr. Larry, whose real name was Larry LeGunn, was not actually a doctor, but a convicted felon whose chiropractic license was revoked in 2010 due to grand theft and insurance fraud.
Dr. Larry wasn’t alone in using strong medications for selling dietary supplements. Jeffrey Bolanos, with a history of drug abuse, led Beamonstar Products in Arizona. Three of the company’s sexual enhancement pills contained tadalafil, a key ingredient in the prescription drug Cialis. These products were eventually recalled by the company.
In 2009, Kilo Sports, a company selling 'natural' performance-enhancing drugs, was found to be selling products containing steroids. In 2010, it was revealed that one of their products also had anti-estrogen substances. Martin McDermott, the head of Kilo, had a criminal record dating back to 2004, with charges related to felony possession of testosterone, boldenone, and human growth hormones. It is believed that McDermott used these substances to illegally enhance the products he sold.
8. Bayer’s HIV Blood Plasma
In 2003, The New York Times conducted an investigation revealing that Cutter Biological, a division of the pharmaceutical giant Bayer, was involved in unethical practices during the 1980s. These practices led to the spread of HIV among hemophiliacs outside of the United States. Allegedly, in 1984, Cutter responded to complaints by creating a safer version of a product, but they continued to sell the hazardous older version in other countries.
The product in question was Factor VIII Concentrate, made from the blood plasma of 10,000 donors to treat hemophiliacs. However, at the time, donor plasma was not tested for HIV due to limited awareness of the disease, which led to hemophiliacs contracting HIV after using the product.
In February 1984, the product was reworked, and the old version was meant to be withdrawn from the market. But company records later showed that the product continued to be sold in countries like Malaysia, Singapore, Indonesia, Japan, and Argentina. Cutter claimed this was due to skepticism about the new drug’s effectiveness and a shortage of plasma, making it difficult to produce the new version.
When distributors in Hong Kong expressed interest in the product in late 1984, Cutter instructed them to use the infected version before the new one. As a result, around 100 hemophiliacs contracted HIV. The continued sale of the infected product in Asia and other developing countries led many customers to accuse Bayer of racial discrimination.
Eventually, Bayer quietly began settling foreign lawsuits linked to the product. After the information became public, Bayer sold its blood plasma business in October 2003.
7. Metal-Tainted Children’s Medication

In 2009, Johnson & Johnson, which produces popular medications like Children’s Tylenol and Children’s Motrin, received reports of black particles in their liquid medicines. These particles, made up of nickel, iron, and chromium, were found in products from McNeil’s plant in Pennsylvania. Investigations revealed the metals were a result of production machinery contamination.
In April 2010, Johnson & Johnson initiated a voluntary recall of the affected liquid medications. This was just one of many recalls linked to McNeil’s plant, with the FDA ultimately attributing the contamination to machinery in the production process.
Despite knowing about the metal contamination, Johnson & Johnson continued to sell the liquid medications for several months after the discovery. Tragically, a child named Joshua Arndt, just four years old, died in 2009 after ingesting a dose of Children’s Tylenol that was contaminated. Doctors could do nothing to save him despite quick emergency treatment.
In 2012, Joshua Arndt’s father took legal action by filing a lawsuit. However, the case was dismissed in 2014 due to the expiration of the two-year statute of limitations. Despite this, Johnson & Johnson faced consequences when, in 2015, charges were filed against them for continuing to sell the harmful medication even after they were aware of its dangers.
Ultimately, Johnson & Johnson agreed to a $25 million settlement to resolve the legal disputes. As for the McNeil plant, the site where the contaminated medication was produced, it was completely torn down and rebuilt.
6. The Deadly Heparin Incident

In 2008, the demand for heparin, a blood thinner, reached alarming levels in the United States, with approximately 300,000 doses required daily. To keep up, producers in China resorted to unethical practices, including sourcing heparin from cow and sheep intestines instead of the usual pig intestines. The FDA reported that this led to the spread of previously unseen diseases.
Doctors identified dangerously low blood pressure as the primary symptom of contaminated heparin, which was observed in hospitals and by patients throughout the United States. By the end of 2008, 81 deaths had already been linked to the drug. The situation worsened as the harmful heparin had passed multiple screenings before reaching the market.
When Baxter International, responsible for distributing half of the U.S. supply of heparin, became aware of the contamination, they immediately issued a recall. Initially, it was believed that only the U.S. had received the tainted drug, but soon it became clear that other countries had been affected. Eleven nations reported similar cases of harm caused by the contaminated heparin.
The contamination was traced to the chemical oversulfated chondroitin sulfate, which is derived from non-pig sources. Although Chinese officials denied that the heparin was tainted, the FDA traced the drug back to 12 Chinese manufacturers. Following this revelation, reforms were implemented in these companies.
Despite the FDA's concerns about Chinese manufacturing practices dating back to 2007, it wasn’t until 2012 that serious regulatory guidelines were established. Since 2007, approximately 246 deaths have been linked to the contaminated heparin, and it is believed that some of the tainted drug may still be circulating in the market nearly a decade later.
5. Contaminated IV Bags
In 2011, 19 individuals at six hospitals in Alabama fell ill due to a specific cause: their IV bags were contaminated with Serratia marcescens bacteremia, a type of bacteria that can be deadly if it enters the bloodstream. Many of the affected patients were already high-risk, receiving intravenous feeding because they were too ill to eat on their own.
The outbreak began in March 2011 at several hospitals in the Birmingham area of Alabama. By the end, 19 people had fallen sick, and nine of them would ultimately die. The bacteria caused immediate effects on the patients, including changes in blood pressure and temperature. However, the surviving patients eventually recovered after receiving treatment.
This wasn’t the first time that infections in IV bags led to patient illness. In Minnesota, a series of painkiller thefts by nurses at local hospitals resulted in infections. At St. Cloud Hospital, patients who were supposed to receive painkillers instead were given saltwater.
This mistake led to rare bacterial infections in 25 patients, six of whom required intensive care, and one who sadly passed away. Blake Zenner, a nurse who stole painkillers between 2010 and 2011, was identified as the source of the outbreak and was finally caught in 2012.
4. Meningitis and Mold in Steroid Injections
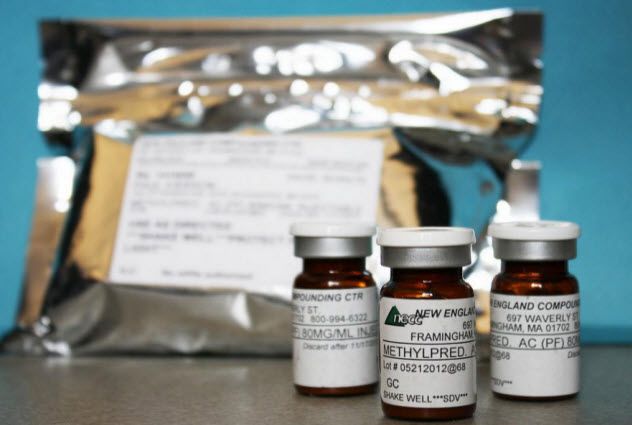
In 2011, the New England Compounding Center began distributing batches of contaminated steroids that carried both meningitis and mold. By the end of the year, the CDC estimated that 14,000 people had been exposed to these infected steroids. The injection of these steroids could result in meningitis, along with a mold that could lie dormant for months before causing harm. The epidemic spread across 16 states, affecting hundreds.
The steroid in question was a methylprednisolone injection, tainted with a rare black fungus known as Exserohilum rostratum, which typically affects plants. It was so uncommon in humans that its incubation period was not recognized at the time of the outbreak. By the end of the year, 268 cases of fungal meningitis, three cases of fungal joint infections, and 21 deaths were linked to the tainted steroid injections.
While most individuals infected with the fungus reported an incubation period ranging from a few weeks to two months, it was also known that the infections could remain dormant for several months. As a result, even after the contaminated steroid injections were recalled, those who had received them could still develop symptoms later.
A similar fungal meningitis outbreak occurred in 2002, involving the same type of steroids. This incident revealed critical lessons about the manufacturing process. It was discovered that without strict standards for mixing the substances, the fungus thrived, leading to both outbreaks.
By 2013, the number of fungal meningitis cases had risen to 751, with 64 deaths. Even a full year after the outbreak, many individuals still required ongoing treatment, highlighting the dangers of neglecting safety protocols in the medical field.
3. Toxic Cough Syrup

This is one of the most tragic cases of contaminated medicine due to its widespread impact and the vulnerability of the people affected: the sick in developing countries. It all began in China, where the chemical glycerine, a primary ingredient in cough syrup, was replaced with diethylene glycol, a sweet-tasting but extremely toxic substance, in order to maximize profits.
Diethylene glycol is an industrial solvent commonly found in antifreeze. When consumed, it leads to kidney failure, paralysis, and ultimately, multiple organ failure.
Decades ago, the use of diethylene glycol in medicines caused over 100 deaths in the United States, prompting the FDA to introduce strict regulations. However, in less developed countries, it continued to be used as a substitute in various syrups and medicines over the next two decades.
This led to at least eight major poisoning incidents, one of which in Panama resulted in 365 reported deaths. A conservative estimate suggests the number of fatalities could be in the thousands. Diethylene glycol was first seen in Bangladesh in 1992 when children died after consuming counterfeit syrups. Later, 88 children died in Haiti.
All of these toxic products trace back to the Yangtze delta, often referred to as “chemical country” by the Chinese due to its reputation for chemical production. Many raw chemicals are manufactured there, and diethylene glycol is frequently sold in place of glycerol.
In numerous cases where widespread fatalities were linked to these products, they underwent several rounds of inspections by distributors. However, the truth remained hidden. Frequently, those producing the chemicals were unlicensed. Driven by the desire to earn a few extra dollars, buyers often chose to ignore the risks.
After purchasing the product, buyers would fraudulently certify it, enabling distributors to accept it. It wasn’t until numerous lives were lost that the Chinese government finally took steps to address the issue. In 2007, the World Health Organization reported that approximately 440 counterfeit operations had been shut down. Hopefully, these actions will help prevent further disasters.
2. Bacteria-Contaminated Ultrasound Gel
In 2011, doctors working in the emergency department at Beaumont Health Center near Detroit began noticing an alarming trend: many patients were testing positive for the bacteria P. aeruginosa. This bacteria is uncommon and usually indicates contamination. Further investigation revealed that all 16 patients had very similar histories, each having developed respiratory illnesses after undergoing cardiovascular surgeries.
Ultrasound gel is used to enhance images during surgeries or medical examinations. Unfortunately, patients experienced illness after this imaging gel was applied to them during surgery, leading to respiratory infections. A 2008 European study revealed that numerous gel bottles tested positive for contamination after bacterial cultures were grown.
At the 18th European Congress of Clinical Microbiology and Infectious Diseases, the researchers presented their findings, alerting that bacteria could potentially infect the gels during production. Despite these warnings, US manufacturers seemingly ignored the advice.
When bacterial cultures were examined from gel bottles used before the outbreak in the United States, they revealed strains of bacteria linked to the manufacturing process. The company responsible for the gel, Pharmaceutical Innovations based in New Jersey, faced a raid by US Marshals, and their gel products were confiscated.
Because imaging gel is widely used, it was uncertain how far the contaminated products had spread. The FDA could only issue a warning to inform everyone using the gel, as bacteria could spread quickly once applied. Thankfully, no further outbreaks occurred, and new safety regulations were soon implemented.
1. Contaminated Syringes: A Silent Threat
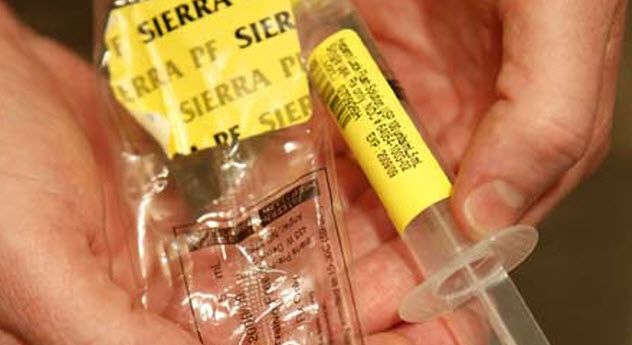
Safety protocols regarding syringes are well known among healthcare providers and patients alike: syringes should never be reused or shared. However, what happens when the manufacturer unintentionally contaminates the syringes? This was the case in 2007 when over 100 individuals became infected with bacteria linked to contaminated saline syringes produced by the same company.
To prevent such outbreaks, companies must implement multiple safety checks. Furthermore, the FDA also reviews medical products. In the case of the contaminated syringes, however, evidence suggests that the FDA missed the contamination.
The prefilled saline syringes were inspected by an FDA official before they were shipped in October 2007. According to the inspection report, the official noticed black, brown, and red particles within the syringes but dismissed them as 'rust' and reported that the factory management had a plan in place to resolve the issue.
The factory had apparently switched to an unreliable sterilization process around the time of the FDA inspection, but this issue was not documented. Just a week later, a distributor pulled 1.3 million syringes from the market, a recall that should have triggered a thorough follow-up inspection from the FDA. However, due to a shortage of staff, they failed to act on it.
When the FDA finally conducted another inspection of the factory, it revealed conditions far below acceptable standards, leading to its closure in January 2008. However, by that point, over 100 people had fallen ill due to the contaminated syringes, with six fatalities. In 2016, B. Braun, the company responsible for the syringes, agreed to a $7.8 million settlement for damages.
