Cloning. IVF. Stem cells. Decades of research into eggs, cells, and embryos have led to extraordinary breakthroughs that have greatly benefited humanity. These scientific strides have enabled remarkable achievements, from overcoming infertility to enhancing everyday life, like improving the quality of beef.
However, scientific progress has also taken some unusual and even unsettling twists, sparking ethical dilemmas along the way. Below are 10 of the strangest phenomena in the field of reproductive science.
10. Baby Hair Lassos and Salamanders

Scientist Hans Spemann discovered that his usual tools were ineffective when working with his test subjects, early-stage salamander embryos. They were simply too slippery. While holding his nine-month-old daughter, an idea struck him. He cut a lock of her hair and returned to the lab.
Using small loops of his daughter’s hair as tools, Spemann conducted his experiments. In one of his first trials, he split the embryos. Contrary to the prevailing theories of the time, the result was independent, fully formed salamanders—artificial twins.
In a subsequent experiment, he squeezed a cell into a dumbbell shape and moved the nucleus to one end of the cell. When the baby-hair noose was released, the nucleus slid back to the other side of the cell, where it divided, essentially creating a clone of the developed side of the embryo.
The results varied depending on where he made the incision and how he applied the constriction. Cutting through one part of the egg led to identical twins, while cutting elsewhere resulted in only one-half of the embryo developing normally, with the other half degenerating into a mass of blood and tissue.
If Spemann slightly constricted the egg, a two-headed salamander formed. The two heads fought over the same body’s food supply, and Spemann referred to them as “two egotisms in the place of one.” He kept these bizarre salamanders for research purposes.
9. Urine Clones
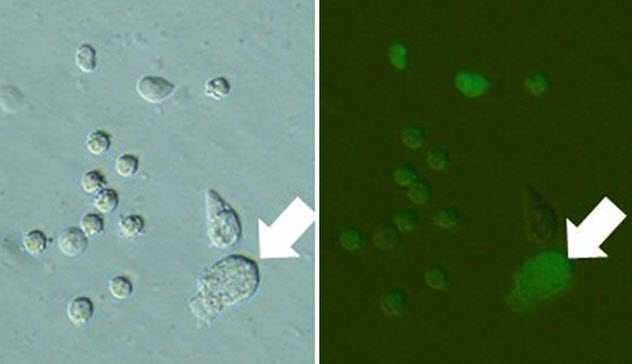
Dolly the sheep was created through a process called nuclear transfer. In this method, the nucleus of an adult sheep’s cell was transferred into an egg cell and then implanted into a sheep to develop.
Nuclear transfer has the potential to aid in the conservation of endangered species. However, obtaining donor cells carries risks, as it could inadvertently harm the animal. Researchers at the University of Yamanashi propose a simpler method to gather donor cells without causing harm: using urine. Urine contains various types of cells, including those from the bladder and kidneys, which can be cultured in the lab after collection.
Generally, urine is not ideal for cell survival. Some of its components are toxic, which led to the belief that even if live cells could be extracted, the environment would likely impair cell survival and the integrity of the nucleus.
But this wasn’t an issue. Some of the urine clones successfully developed to an early embryonic stage and were then transferred to surrogates for further growth. When these urine clones reached maturity, they were able to reproduce with each other. The researchers' tests on their fertility strongly indicate that cells from urine remain viable for cloning purposes.
However, there remains a challenge. Collecting urine cells from wild animals is limited, especially under clean conditions.
8. IVF Through the Ages
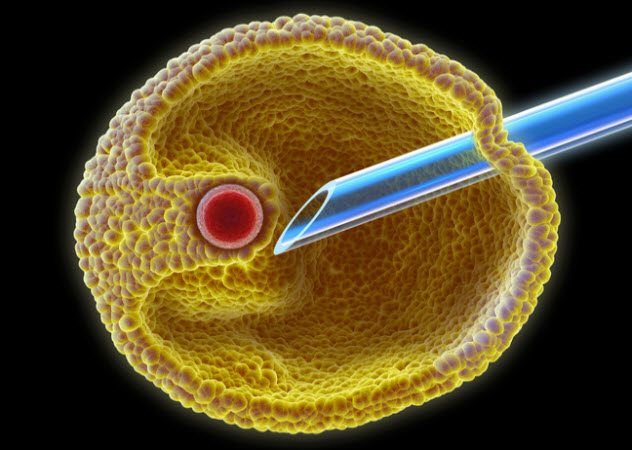
In vitro fertilization (IVF) has given infertile couples the opportunity to bypass their biological limitations and have children. In this process, gametes are placed in a small glass dish where they merge to form a zygote. Once the zygote progresses to the early embryo stage, it is implanted back into a woman's body to continue developing. IVF is now a routine (though costly) procedure, and millions of babies have been born through it.
Yet, just a few decades ago, IVF was considered both unethical and unattainable, with the scientists behind the first IVF baby even being accused of “playing God.” Of course, the process has been refined significantly since those early days.
In the early days of IVF, the process was far more demanding and shrouded in secrecy due to the controversy surrounding it. Women were required to stay as inpatients at the clinic for two to three weeks, residing in portable buildings on the clinic grounds. They had to collect all their urine during treatment, as this was the only method by which doctors could track hormone levels. Patients had to provide samples every three hours, even at night.
At one point, eggs had to be collected via keyhole surgery, which involved making small incisions in the body. Today, a newer, safer technique is used that only requires mild sedation. This modern method allows eggs to be extracted through a needle under ultrasound guidance, and the entire process takes about 30 minutes.
7. Cloning Anomalies
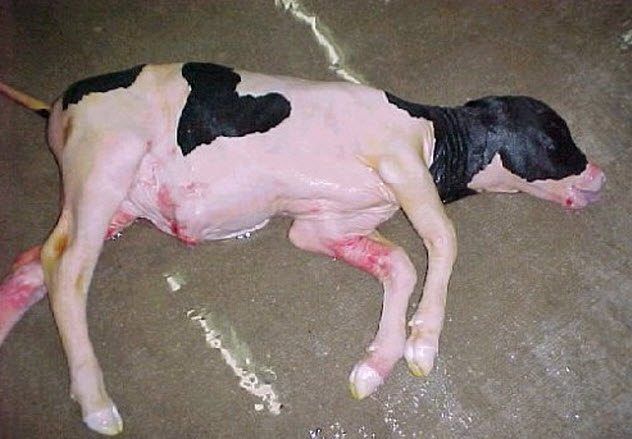
Dolly the sheep, celebrated as the first successful clone of an adult mammal, remains the lone success in a long series of failures. Out of 277 clones created by the scientists who produced Dolly, only she survived to be born.
Failed pregnancies and deaths are a natural part of reproduction. Stillbirths and birth defects can occur regardless of how an animal is conceived. Nevertheless, cloning has a long history of complications.
Some abnormal fetuses manage to reach full term, only to exhibit abnormalities at birth. One of the most common conditions in clones is “large offspring syndrome.” Calves or lambs affected by this syndrome are 30–40 percent larger than normal, making birth difficult. Additional health issues may affect organs such as the brain, heart, and liver.
Cloned calves and lambs with birth abnormalities may experience health complications during the early months of life. However, after six months, these animals show no noticeable differences in appearance or blood measurements when compared to conventionally bred animals of the same age.
The welfare concerns raised by cloning led to the European Parliament's ban on farm animal cloning in 2015. In contrast, cattle cloning continues in the United States, with hundreds of cases each year.
6. Resurrection From A Steak

The challenge with cloning cows to produce the finest beef is that it’s impossible to determine the meat’s quality without slaughtering the cow. Thankfully, there's a workaround: simply clone from the steaks.
Only 3 out of every 10,000 cow carcasses qualify as the ideal rib-eye steak, which is marbled with fat for flavor but lacks the unwanted thin layer of back fat. When a scientist at West Texas A&M University (WTAMU) saw two such rare rib-eyes pass by in quick succession, he contacted Dean Hawkins, the head of the animal sciences department at the university.
A small amount of meat was sufficient to grow cells, extract DNA, and insert it into an egg cell from a cow. One steak came from a castrated bull, and the other came from a cow that had never given birth. Four clones were created from these two steaks. One clone, named Alpha, came from the bull, while three clones, named Gamma 1, Gamma 2, and Gamma 3, came from the female cow.
WTAMU bred the clones to pass on the genes for top-quality beef. The 13 calves born from these clones were the first bovine offspring produced from two cloned carcasses. Seven of these calves were later slaughtered. The evaluation of their carcasses showed they were graded significantly above the industry average.
5. ET and Cows

In the 1970s, American ranchers began challenging nature’s constraints with a technique known as embryo transfer (ET). While a cow typically carries just one embryo at a time, cows undergoing ET therapy can produce six or seven viable embryos, with some even capable of producing 80 to 90 embryos simultaneously.
These embryos are extracted using thin tubes and then implanted into surrogate cows to carry them to term. Thanks to ET, farmers can generate dozens of calves annually from genetically superior cows without those cows ever needing to give birth themselves.
However, ET poses a risk of inbreeding. If farmers choose to breed exclusively from the offspring of a single cow, it could reduce the genetic diversity of the herd, making it more susceptible to disease. Interestingly, ET can also aid in genetic diversity. The USDA has accumulated a stockpile of embryos from various livestock breeds at Fort Collins, Colorado.
4. Artificial Twinning

In a sense, clones are quite natural. When a single fertilized egg divides into two, creating two genetically identical individuals, the result is simply identical twins. The easiest way to clone an animal is to split an early-stage embryo, resulting in artificial twins. This technique has been widely used in cows, producing thousands of calves.
The first experiment in artificial twinning was conducted by Hans Driesch in 1885. Driesch shook a container holding a two-cell sea urchin embryo, separating the cells. Both cells then developed into healthy sea urchin larvae. In 1902, Hans Spemann replicated this with vertebrates by using a baby hair noose to split a salamander embryo.
In 2000, a rhesus monkey named Tetra became the first primate cloned through artificial twinning. Tetra was created by splitting an eight-cell embryo into four two-cell pieces.
Although the technique varied, Tetra’s creation was similar to Dolly’s in that it required many attempts. The researchers produced 368 embryos by splitting 107 embryos into two or four parts. Not all the embryos survived. Among all the surrogate mothers, only Tetra’s surrogate had a successful pregnancy.
3. The Story of the Mouse Princess
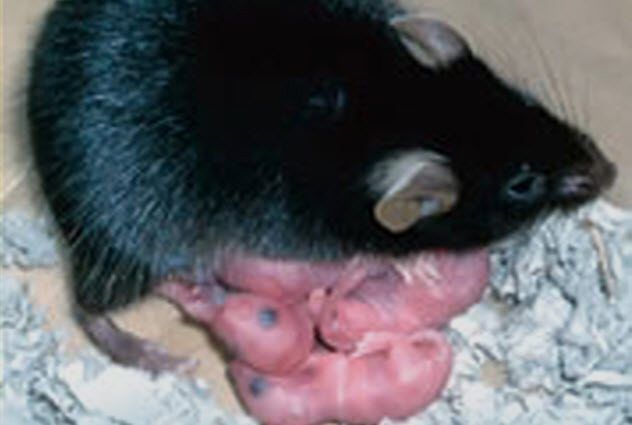
In 2004, researchers at the Tokyo University of Agriculture made a remarkable breakthrough, creating a mouse without a father—a feat that seemed like something out of a fairy tale.
While it's possible to artificially stimulate mammal egg cells to divide, the embryos produced typically fail to develop properly and perish in the womb. Scientists had long suspected this outcome was related to a process called imprinting, which involves certain genes being switched on or off during the formation of gametes.
To bypass this obstacle, the scientists fused an immature egg cell, which hadn't undergone imprinting, with a mature one. The immature egg was taken from a genetically modified mouse that lacked genes believed to interfere with the development of unfertilized eggs into viable embryos.
This happy ending was hard-earned. Out of 457 egg fusions, only 371 survived to the early stages of embryonic development before being transferred to surrogate mothers. Just 10 live mouse pups were born, and only one made it to adulthood. This sole survivor was named Kaguya, after a Japanese fairy-tale princess who was discovered as a baby inside a bamboo stalk.
Scientists caution that it's too early to consider applying this technique to humans. The method is highly labor-intensive, and its failure rate is significant. Moreover, it would require genetically altering human eggs, which is widely viewed as ethically unacceptable.
2. Artificial Embryos
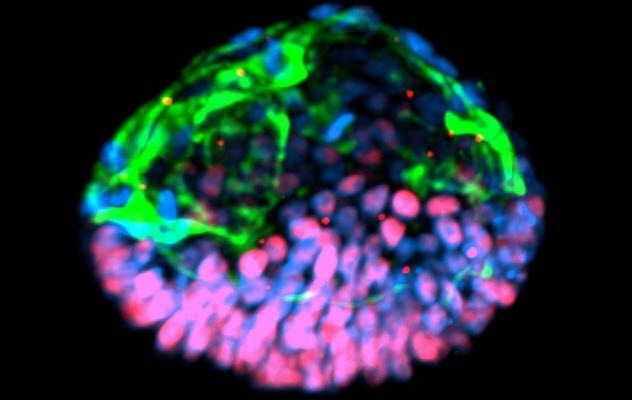
Researchers from the University of Cambridge announced they had successfully created an artificial mouse embryo using two distinct types of stem cells. One of the types was embryonic stem cells, while the other typically forms the placenta.
The scientists placed the stem cell mixture onto a 3-D scaffold designed to imitate the natural cell support structure, aiding in the proper development of the growing cells. After four and a half days, the cells had developed in such a way that they resembled a normal mouse embryo.
Magdalena Zernicka-Goetz, the lead author of the study, explained to Reuters that this method will allow researchers to study key stages of human development without directly working on embryos. She also mentioned that understanding the typical course of development will provide insight into why it often goes wrong.
The proposal brings up ethical concerns. At present, scientists are only permitted to use discarded human embryos, and they can only keep them alive for up to 14 days after fertilization.
1. Embryo Screening

Preimplantation genetic diagnosis (PGD) is a form of embryo screening used to help couples undergoing in vitro fertilization (IVF) avoid passing on genetic mutations that could lead to disabilities or diseases in their children. PGD typically involves removing one or a few cells from an IVF embryo to analyze its gene sequences and chromosomes. Embryos that do not meet the criteria are usually discarded.
Fertility clinics in China are rapidly expanding. The largest clinic recorded 41,000 IVF procedures in 2016, which accounts for about a quarter of the total number of procedures performed in the United States annually. With preimplantation genetic diagnosis (PGD) procedures growing by an estimated 60–70 percent each year, China is expected to match the US in per capita procedures within the next few years.
Efforts to eliminate genetic disorders and disabilities raise significant ethical concerns. Some people fear that the drive to eradicate disabilities undermines the value of the lives of those already living with them. Additionally, the cost of PGD could exacerbate existing social inequalities, with wealthier individuals having greater access to these procedures. However, in China, most of the focus is on the benefits of the procedure.
Although clinics authorized to perform PGD are only allowed to use it for preventing serious diseases or aiding infertility treatments, some patients request more. Sijia Lu, the chief technology officer at Yikon Genomics, reports that some families have asked for the procedure to eliminate a genetic mutation that prevents many Asians from processing alcohol, a condition that can complicate participation in alcohol-heavy business lunches in China. (The company has declined such requests.)
