 The intricate design of the chloroplast. | Encyclopaedia Britannica/Getty Images
The intricate design of the chloroplast. | Encyclopaedia Britannica/Getty ImagesYou’ve heard of the sun, right? That immense fiery sphere radiates vast amounts of energy, fueling every living thing on Earth, beginning with the plants. The sun releases various types of electromagnetic radiation, and plants utilize the visible light to power the incredible process of photosynthesis.
Although photosynthesis may seem like a mystical phenomenon, it's actually a complex chemical process carried out by chloroplasts — tiny organelles found in plants and eukaryotic algae (eukaryotic meaning having a well-defined nucleus) — that trap sunlight and transform that energy into food for the plant.
Chloroplasts Originated from Ancient Bacteria
Chloroplasts function similarly to mitochondria, another organelle found in eukaryotic cells that also generates energy. This resemblance makes sense since both originated from a long-ago bacterium that was engulfed by, but not consumed by, a larger cell. This led to a unique partnership between two organisms, explained by the "endosymbiont hypothesis." Both chloroplasts and mitochondria have their own DNA and reproduce independently of the cell.
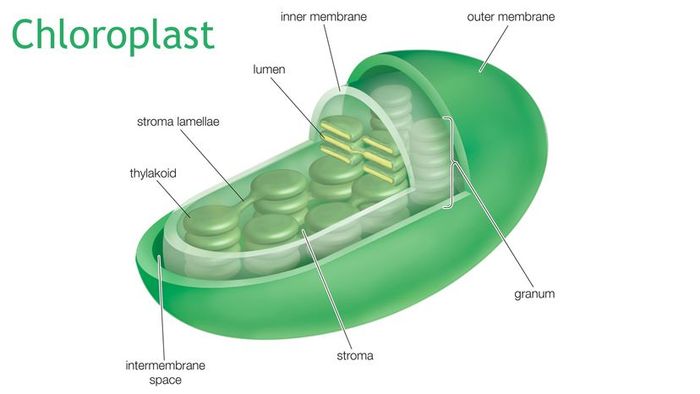
Chloroplasts are present in all green parts of the plant. They consist of a double membrane, with the inner part containing tiny pockets (called thylakoids) filled with a pigment known as chlorophyll. These thylakoids are suspended in a fluid called stroma.
The secret to the chloroplast's photosynthetic power lies in its membranes. Having originated as an independent bacterium, chloroplasts retain two membranes: the outer membrane is from the engulfing cell, while the inner membrane is the bacterium’s original one. Imagine the outer membrane as the wrapping paper and the inner one as the box the toy came in. The crucial area for photosynthesis is the space between these two membranes — where the thylakoids reside.
Chloroplasts Operate on Gradients, Much Like Batteries
The chloroplast's double membrane creates four distinct compartments: the area outside the cell, the cytoplasm within the cell, the stroma inside the chloroplast but outside the thylakoids (essentially the space between the inner and outer membranes), and the thylakoid space, which corresponds to the original bacterial cell. The thylakoids are small, membrane-bound stacks of pouches that act as dividers, preventing random movement of particles between compartments. This structure allows the chloroplast to concentrate charged particles in specific areas and move them across spaces through controlled channels.
"That’s how batteries function," explains Brandon Jackson, an associate professor in the Department of Biological and Environmental Sciences at Longwood University in Farmville, Virginia. "It requires energy to gather a lot of negative electrons at one end of the battery and a lot of positive charges at the other. When you connect the two ends with a wire, the electrons eagerly flow to balance the electrochemical gradient. If you place something along that wire — like a light bulb, motor, or computer chip — the electrons will travel through, doing useful work. If not, the movement will still release energy, but as heat."
Jackson further explains that to create a battery within a plant cell, an energy source and dividers are needed to establish and maintain gradients. If the gradient collapses, some of the energy used to create it is lost. In the case of the chloroplast battery, this electrochemical gradient is generated when the plant absorbs solar energy, and the thylakoid membranes act as dividers, separating different concentrations of hydrogen ions (protons) that have been stripped from water molecules.
Trace the Energy
Within the chloroplast, complex chemical reactions are taking place, but the outcome is simple: sunlight is transformed into stored energy, essentially creating a biological battery.
So, let's follow the energy:
The sun shines on a leaf. That solar energy excites electrons inside water molecules in the leaf, and because excited electrons bounce around a lot, the hydrogen and oxygen atoms in the water molecules break apart, launching these excited electrons into the first stage of photosynthesis — a conglomeration of enzymes, proteins and pigments called photosystem II, which breaks down water, producing hydrogen ions (protons that will be used in the battery and oxygen gas that will float off into the air as plant garbage).
These energized electrons get passed on to some other membrane-bound proteins that use that energy to power ion pumps that escort the hydrogen ions from the space between the membranes into the thylakoid space, which is where all the light-dependent reactions of photosynthesis occur. Photosystems and electron pumps cover the surfaces of the thylakoid membranes, pumping the hydrogen ions from the stroma (the fluid space between the thylakoid and the inner membrane) into the stacks and stacks of thylakoid pouches — and these ions really want to get out of these thylakoids, which is what creates the electrochemical gradient. In this way light energy — that stuff that shines on your face when you go outside — is converted into a sort of battery, like the ones that run your wireless earbuds.
At this point, photosystem I takes over, which arranges for temporary storage of the energy generated by the battery. Now that the electron has been allowed to move along the gradient, it's much more relaxed, so it absorbs some light to re-energize it, and passes that energy along to a special enzyme that uses it, the electron itself, and a spare proton to make NADPH, which is an energy-carrying molecule that provides short-term storage for chemical energy that will later be used to make glucose.
At this point, the light energy is now in two places: It's stored in the NADPH and as the electro-chemical gradient of the difference in hydrogen ion concentration inside the thylakoid compared to just outside it in the stroma.
"The high hydrogen ion gradient inside the thylakoid wants to break down — it must break down," explains Jackson. "Gradients are a form of 'organization,' which is essentially the opposite of entropy. Thermodynamics tells us that entropy will always seek to increase, meaning the gradient must dissolve. The hydrogen ions inside each thylakoid want to escape to balance the concentration across the inner membrane. However, charged particles can't move freely through a phospholipid bilayer — they require a specific channel, just as electrons need a wire to travel from one side of a battery to the other."
Similar to how you can attach an electric motor to a wire to power a car, the channel through which hydrogen ions pass works like a motor. These protons flow through their designated channel, much like water flowing through a hydroelectric dam down an elevation gradient. This movement generates enough energy to trigger a reaction that creates ATP, another form of short-term energy storage.
At this point, the light energy has been converted into short-term chemical energy in the form of NADPH and ATP. These molecules will be useful later in the dark reactions (also known as the Calvin Cycle or carbon-fixation cycle) inside the chloroplast. This process occurs in the stroma, where enzymes convert NADPH, ATP, and carbon dioxide into sugars that either nourish the plant, aid respiration, or help produce cellulose.
"Building complex organic molecules like cellulose, which is made of glucose, takes considerable energy — all derived from the sun," says Jackson. "This energy journey starts as light wave energy, then moves to excited electron energy, followed by electrochemical gradient energy, and finally chemical energy in the form of NADPH and ATP. Oxygen gas is released, and instead of the NADPH and ATP being used for other cellular tasks, they are transferred to the carbon-fixation cycle, where enzymes break them down, harness the energy, and use it to synthesize glucose and other organic molecules."
And all of this, thanks to a tiny organelle called the chloroplast.
Since chlorophyll absorbs red and blue light effectively, but reflects green light, leaves appear green to our eyes because that is the color of light that is reflected off them.
