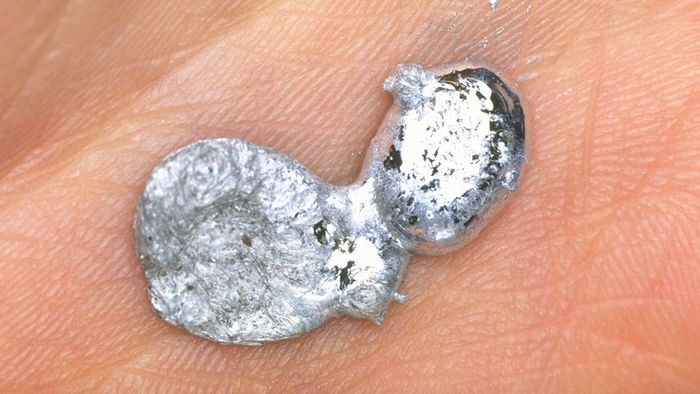
 Gallium, a chemical element with a melting point of 85.6°F, melts when held in your hand. Lester V. Bergman/Getty Images
Gallium, a chemical element with a melting point of 85.6°F, melts when held in your hand. Lester V. Bergman/Getty ImagesGallium is a rare, silvery-white metal capable of performing one of the most fascinating tricks on the periodic table. At room temperature, it appears as a shiny metallic solid, similar to pure aluminum. But just a few minutes in your hands and this solid metal starts to turn into liquid.
Indeed, gallium's melting point is a mere 85.6°F (29.8°C), meaning it transforms into a reflective puddle in your warm hand. In its liquid state, gallium resembles mercury, but it is non-toxic and safer to handle, although it can stain the skin.
Gallium isn't just a metal that melts in your hand for viral YouTube videos. It's also crucial in LED technology and is the preferred semiconductor for the high-performance microchips found in smartphones. The only thing holding gallium back from dominating the electronics industry is its rarity and high cost compared to silicon.
Mendeleev Predicted the Existence of Gallium
Pure gallium doesn't occur naturally in its shiny, elemental form. It has to be extracted from minerals like bauxite via a complex chemical process. As the U.S. Geological Survey reports, gallium is present in Earth's crust in only 19 parts per million, a stark contrast to silicon, which has 282,000 parts per million. The first scientist to isolate and identify gallium as a new element was French chemist Paul-Emile Lecoq de Boisbaudran in 1875. He named it after the Latin word for France, "Gallia."
Four years before Boisbaudran's discovery, the renowned Russian chemist Dmitri Mendeleev had already predicted gallium's existence. Mendeleev, widely regarded as the "father of the periodic table," noticed a gap in the table following aluminum and theorized that an element he called "eka-aluminum" would share many of aluminum's properties but possess a distinct atomic structure.
Mendeleev's prediction was correct, but he couldn't have foreseen how gallium's unique characteristics — a mix between metal and nonmetal — would make it a perfect fit for modern electronics.
An Element With an Identity Crisis
Here’s another fascinating and somewhat odd fact about gallium: It melts at a relatively low 85.6°F (29.8°C), but it doesn't boil until it reaches a blistering 3,999°F (2,204°C). This unique property gives gallium the distinction of having the longest liquid phase of any element. But what’s the reason behind this?
"Gallium is conflicted," says Daniel Mindiola, a chemistry professor at the University of Pennsylvania, whom we contacted through the American Chemical Society. "It melts at a low temperature, typical of a light element, but boils at a much higher temperature, characteristic of a heavy element. Gallium can't decide if it wants to be a metal or a nonmetal."
Gallium’s dual nature can be traced to its position on the periodic table, where it straddles two categories: "metalloids" and "post-transition metals." Gallium comes right after aluminum, but its atoms are far more "independent" than those of aluminum, which is more "electropositive," a hallmark of true metals, says Mindiola.
Much like silicon, gallium is a decent conductor of electricity, but not the best. This makes both elements ideal candidates for use in semiconductors, where the regulation of electrical flow is crucial.
"Gallium is actually a superior semiconducting material compared to silicon," explains Mindiola. "The challenge is its rarity, which drives up its cost."
When using current manufacturing techniques, a gallium arsenide wafer—the most common gallium-based semiconductor—is approximately 1,000 times more costly than a silicon wafer.
 Gallium arsenide plays a key role in the silicon chips used for radar assistance systems in vehicles.
image credit: picture alliance/Getty Images
Gallium arsenide plays a key role in the silicon chips used for radar assistance systems in vehicles.
image credit: picture alliance/Getty ImagesGallium is Present in Your Gadgets
Although gallium is significantly more expensive than silicon, it has become a popular choice for semiconductors in the latest smartphone models. Smartphones rely on radio frequency (RF) chips to connect with cellular data networks, and RF chips made from gallium arsenide produce less heat compared to silicon while being capable of operating at higher frequency bands, which are essential for 5G connectivity. Over 70 percent of all gallium consumed in the U.S. is used in making RF chips and other integrated circuits, according to the USGS.
One of the most exciting uses of gallium is in light-emitting diodes (LEDs), which are now found in everything from computer screens to traffic signals to high-end car headlights. LEDs are favored for their efficiency in converting electricity directly into light. The first visible-light LEDs were created in the early 1960s when General Electric researchers uncovered the unique properties of diodes made from various gallium alloys (combinations of gallium, arsenic, nitrogen, phosphorus, and other elements).
In a diode, electrons travel through two layers of semiconductor material, one positively charged and the other negatively charged. As free electrons from the negative side fill the "holes" in the positive side, they release a photon of light as a byproduct. Scientists have discovered that different gallium alloys produce photons with varying visible light frequencies. Gallium arsenide and gallium phosphide emit red, orange, and yellow light, while gallium nitride emits blue light.
"All you need to do is apply a current to an LED and it lights up like a Christmas tree," remarks Mindiola.
LEDs not only emit light when connected to electricity, but the process works in reverse as well. The specialized diodes used in solar cells are also made from gallium-based semiconductors. These diodes capture incoming light and separate it into free electrons and "holes," creating voltage that can be stored in a battery as electricity.
Other Fascinating Applications of Gallium
"Medical science is also exploring the uses of gallium in diagnosing and treating specific cancers," explains Mindiola. "Gallium-67 targets cells that grow faster than normal, like those in a tumor."
Gallium-67, a radioactive isotope of gallium, emits harmless gamma rays. By injecting this isotope into a patient's bloodstream, radiologists can conduct whole-body scans to detect tumors or infections. The isotope binds to clusters of rapidly dividing cells, revealing these potential problems in PET scans or other gamma-sensitive scans. Gallium nitrate has also proven useful in shrinking and eliminating certain tumors, not only for detection but also treatment.
For decades, the aerospace sector has been invested in gallium. The high-efficiency solar panels used to power satellites and long-range space missions, including those on the Mars Exploration Rovers, are constructed from gallium arsenide. These gallium-based solar cells could generate up to 900 watt-hours of power per Martian day when operating at full capacity.
By combining specific amounts of gallium, indium, and tin, you create a metal alloy that melts at -2.2°F (-19°C). This liquid metal, sold under the brand name Galistan, serves as a non-toxic alternative to mercury in thermometers.
