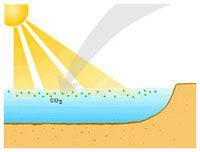
 Phytoplankton utilize CO2 and sunlight for energy through photosynthesis.
Image courtesy NOAA
Phytoplankton utilize CO2 and sunlight for energy through photosynthesis.
Image courtesy NOAAGlobal warming has emerged as a key concern in the 21st century. As scientists warn of increasingly dire consequences, such as Florida being submerged in the coming decades, the public is pushing for immediate action.
Enter visionary scientists and companies like Planktos and Climos, who propose introducing iron into the world's oceans to lower atmospheric carbon dioxide levels, which could ultimately cool the planet. The concept of iron dumping to reduce temperatures dates back to the late 1980s and is referred to as carbon sinking, ocean seeding, or iron fertilization.
The concept is quite straightforward. Iron serves as a nutrient for many plants, and certain ones, like phytoplankton that form the foundation of the marine food chain, require it for growth. By adding iron to the water, phytoplankton flourish, absorbing CO2 during photosynthesis. This leads to a reduction in CO2, which is expected to help lower temperatures since CO2 is one of the main gases that trap heat on Earth's surface through the greenhouse effect.
Since oceanographer John Martin proposed the idea over 15 years ago, a number of iron fertilization trials have been carried out [source: Haiken]. A 2004 experiment showed that every atom of iron introduced into the water could pull between 10,000 and 100,000 carbon atoms from the atmosphere by stimulating plankton growth [source: Schiermeier]. Some scientists speculate that introducing iron into the Southern Ocean could cut carbon dioxide levels by 15 percent [source: Schiermeier].
Scientist Oliver Wingenter advocates for a more cautious approach, warning that large-scale iron addition to the ocean could lead to a drastic cooling of more than 10 degrees Celsius [source: Wingenter]. He proposes fertilizing just 2 percent of the Southern Ocean to achieve a 2-degree Celsius drop in temperature and delay the tipping point of global warming by 10 or more years [source: Wingenter].
Rather than focusing solely on reducing carbon dioxide levels, Wingenter’s research has focused on boosting other gases released by phytoplankton blooms, particularly dimethyl sulfide, or DMS. DMS plays a significant role in cloud formation in polar regions and could increase cloud reflectivity, thereby cooling the atmosphere. In his iron fertilization experiments, Wingenter observed a five-fold increase in DMS concentrations [source: Wingenter].
Other scientists, however, caution against using the ocean as an experimental site. To understand their concerns, read more on the next page.
Concerns About Iron Fertilization
 In this satellite image, the two central green cloud-like formations represent significant phytoplankton blooms. These masses are crucial to understanding oceanic biology.
Photo courtesy of NASA.
In this satellite image, the two central green cloud-like formations represent significant phytoplankton blooms. These masses are crucial to understanding oceanic biology.
Photo courtesy of NASA.It's possible that adding iron to the oceans might have effects beyond simply lowering carbon dioxide levels. By introducing iron and boosting phytoplankton growth, the ocean's food web could be altered in such a way that it would change the balance of various gases in both the atmosphere and ocean. In some cases, these changes could even counteract the benefits of carbon dioxide reduction. For instance, some computer simulations suggest that the addition of iron might lead to higher concentrations of nitrous oxide and methane—two potent greenhouse gases. Mark Lawrence, a scientist in the field, also notes that phytoplankton blooms produce methyl halides, which can deplete the ozone layer.
There's still uncertainty about how long carbon dioxide captured by phytoplankton remains stored in the ocean. While the ocean can accumulate millions of tons of carbon dioxide for over a century, the key factor is that it must sink beneath the surface to stay in solid form. Unfortunately, this does not always happen.
Ocean currents can easily bring the carbon dioxide back up to the surface, releasing it into the atmosphere once again. In fact, up to 95 percent of the carbon dioxide stays in the ocean's circulation, being constantly recycled through the food chain [Wright]. Plankton absorb it, only to be consumed by other organisms that exhale the carbon dioxide back into the atmosphere, preventing it from ever reaching the ocean floor [source: Haiken].
Many environmental groups oppose the practice of ocean seeding. When Planktos, a company, announced its plan to distribute 100 tons of iron sulfate off the coast of the Galapagos Islands, several organizations, including the Natural Resources Defense Council, the World Wildlife Fund, Friends of the Earth, and Greenpeace, voiced strong opposition. One of these groups even threatened to intercept the company's ship. Although Planktos ultimately canceled its plans due to lack of funding and proper equipment, environmental groups continue to keep a watchful eye. On its website, the World Wildlife Fund highlights several potential harmful consequences of this method.
- Phytoplankton blooms can release a significant volume of gases, and the bacteria that form as the plankton decompose will also contribute to this emission.
- As the plankton decay, the bacterial activity will consume oxygen in the water, which could lead to higher levels of gases such as nitrous oxide.
- If the iron used in fertilization isn't pure, it may also bring along other potentially harmful trace metals.
Iron fertilization raises intriguing questions. The interaction between the carbon dioxide cycle and oceanic processes is intricate, and the long-term impact of altering this balance remains uncertain. Is iron fertilization a solution for global warming or a Pandora's box waiting to be opened? Only time will provide an answer, but researchers will undoubtedly continue to seek the truth. To dive deeper into global warming and explore potential carbon sequestration methods, be sure to check out the resources on the next page.
