 Expedition Four flight engineer, Astronaut Daniel W. Bursch, is seen performing maintenance on the Elektron Oxygen Generator within the Zvezda Service Module on the International Space Station (ISS).
Image courtesy of NASA
Expedition Four flight engineer, Astronaut Daniel W. Bursch, is seen performing maintenance on the Elektron Oxygen Generator within the Zvezda Service Module on the International Space Station (ISS).
Image courtesy of NASAOn Earth, fresh air is abundant. Humans inhale oxygen and exhale carbon dioxide, which plants then convert back into oxygen through photosynthesis. This natural cycle operates on a massive scale. But how does this process work within the cramped quarters of spacecraft, such as the space shuttle or space stations?
Spacecraft typically carry their own oxygen supply, often supplemented by a backup system. These missions are usually short, ranging from days to two weeks. However, the International Space Station (ISS), designed for extended missions, has been operational since 1998. So, how does the ISS generate oxygen? It employs one of three methods: oxygen generators, pressurized oxygen tanks, or solid fuel oxygen generators (also known as oxygen candles).
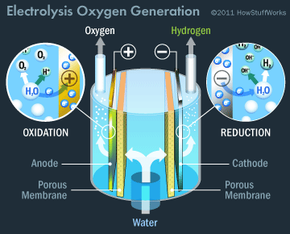
The main approach involves oxygen generators: the Russian-designed Elektron and the U.S.-built Environmental Control and Life Support System (ECLSS). The Elektron is housed in the Zvezda service module, while the ECLSS is situated in the Destiny laboratory module. These systems produce oxygen from water through electrolysis, a process where an electric current flows through water from a positively charged anode to a negatively charged cathode. A small amount of salt is added to the water to enhance conductivity, as pure water is a poor conductor. This process splits water into hydrogen and oxygen gases. Here's the chemical breakdown:
- At the cathode, a reduction reaction takes place. Electrons (e) from the cathode interact with water (H2O) to produce hydrogen gas (H2) and hydroxide ions (OH): 2H2O (l) + 2e ->H2 (g) + 2 OH (aq).
- At the anode, an oxidation reaction occurs. Electrons are extracted from water and flow into the anode, resulting in oxygen gas (O2) and hydrogen ions (H): 2H2O (l) -> O2 (g) + 4 e + 4 H
The station's solar panels generate electricity, which powers the oxygen generators via the station's power grid. Water is transported to the station by Progress supply ships and the space shuttle. Additionally, condensers extract water vapor from the cabin air (exhaled by astronauts), and the ECLSS unit recycles water from astronauts' urine. The hydrogen gas produced during electrolysis is released into space, while the oxygen gas is circulated into the cabin air.
Now, let's explore the alternative methods the ISS uses to generate oxygen.
Creating Oxygen Aboard the ISS
 Electrolysis of water
HSW
Electrolysis of water
HSWAs previously discussed, oxygen on the International Space Station (ISS) is generated through three primary methods: oxygen generators, pressurized oxygen tanks, or solid fuel oxygen generators. We've already covered oxygen generators. Now, let's delve into the remaining two approaches.
The second method involves transporting oxygen to the ISS from Earth. When Progress supply ships, European automated transfer vehicles, or the U.S. space shuttle dock with the station, they transfer oxygen into pressurized tanks located at the airlock nodes. Nitrogen gas is also pumped into separate pressurized tanks at these airlocks. The station's atmospheric systems then blend these gases to match Earth's atmospheric composition and distribute the mixture throughout the cabin.
The third method serves as a backup, generating oxygen through chemical reactions. This system, known as the solid fuel oxygen generator (SFOG), is housed in the Zvezda service module. Often referred to as oxygen candles or chlorate candles, the SFOG contains canisters filled with a mixture of powdered sodium chlorate (NaClO3) and iron (Fe) powder. When activated, the iron combusts at 1112 degrees F (600 degrees C), providing the necessary heat for the reaction. The sodium chlorate decomposes into sodium chloride (table salt - NaCl) and oxygen gas (O2). Some of the oxygen reacts with iron to form iron oxide (FeO):
1112°F
NaClO3 (s) + Fe (s) -> 3O2 (g) + NaCl (s) + FeO (s)
Each kilogram of the SFOG mixture provides 6.5 man-hours of oxygen. Additionally, Russian spacesuits utilize SFOGs to generate oxygen.
For future space stations or colonies, NASA researchers aim to produce oxygen and remove carbon dioxide naturally by cultivating plants. These plants would not only provide breathable air but also serve as a food source for astronauts. However, a key challenge is developing methods to grow large quantities of plants in confined spaces, as living areas on space stations are limited.
