 Can we create cloned organs that are perfect genetic matches?
Can we create cloned organs that are perfect genetic matches?Imagine having a clone of yourself stored away just in case you ever needed a new heart or liver, like a spare tire in your car's trunk. This idea was central to the 2005 low-budget sci-fi film, "The Island," and also ties into the concept of therapeutic cloning.
In the movie, Hollywood stars Scarlett Johansson and Ewan McGregor play dual roles as wealthy individuals and their genetically identical clones. In a chilling Orwellian twist, doctors are forced to kill the 'spare' clones to harvest their organs.
While "The Island" isn't likely a realistic portrayal of the future of human cloning, it raises important questions about the practical and economic implications of both reproductive and therapeutic cloning.
Challenges in Organ Transplantation
Organ transplants are complex and challenging for two key reasons. The first hurdle is finding a suitable donor, and the second is the uncertainty of whether your immune system will accept the new organ.
The gap between organ demand and availability is staggering. In the first half of 2023 alone, 31,121 Americans underwent organ transplants, with around 85 percent of those coming from deceased donors. However, as of September 2023, over 103,425 individuals were on the national waiting list for organs [source: OPTN].
Potential of Embryonic Stem Cells
Imagine eliminating both the waiting period and the uncertainties that come with traditional organ transplants by creating personalized, cloned organs from your own cells that your body would naturally accept.
Cloning proponents refer to this field of study as therapeutic cloning. This differs from reproductive cloning because therapeutic cloning is focused solely on embryos and does not involve bringing human babies to full term.
Embryos house pluripotent embryonic stem cells, which are capable of differentiating into over 200 distinct types of cells. These stem cells are extracted when embryos reach the blastocyst stage, a phase in which the embryo is composed of roughly 150 cells.
However, extracting these stem cells results in the destruction of the embryo. This practice of terminating embryos through cloning has sparked significant controversy, with many both within and outside the scientific community opposing its use, contributing to an ongoing debate on the ethics of human embryonic stem cells.
Setting the controversy aside, how would cloned organ transplants work? Clearly, doctors cannot simply remove your heart and create an identical one on the spot. Additionally, cloning yourself to harvest organs wouldn't be a viable solution. This is where stem cells come into play, along with recent breakthroughs in science that bypass the need for cloning altogether.
How Organ Cloning Could Work
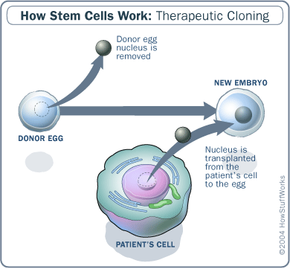
To grasp the concept of organ cloning, it's important to first understand what cloning entails. One of the most prevalent techniques for both therapeutic and reproductive cloning is somatic cell nuclear transfer (SCNT). This method involves extracting the nucleus from a donor egg and replacing it with the DNA of the organism that is to be cloned.
Cloning organs using somatic cell nuclear transfer could potentially be done by cloning human embryos, extracting stem cells from the blastocyst, and guiding these stem cells to form the organ of interest. For instance, generating a liver from human stem cells will require more extensive research. Scientists aim to reverse-engineer cell differentiation processes to identify the specific chemical or physical signals that prompt stem cells to differentiate correctly.
Human therapeutic cloning research has largely slowed in the United States due to bioethical concerns and a shortage of available human eggs for research. Ethical guidelines from the National Academy of Sciences and the International Society for Stem Cell Research prohibit paying women for donating eggs for embryonic stem cell studies. [source: NCBI]
The relatively new field of stem cell research, combined with the risks associated with egg donation, has made it challenging for scientists to find willing donors. Since embryonic cloning often has low success rates, researchers require a large number of eggs to make meaningful progress. To address the shortage of human eggs, Ian Wilmut, the scientist who cloned Dolly the sheep, has proposed injecting human DNA into animal eggs. [source: Britannica]
Despite obstacles, there have been significant strides in therapeutic cloning through animal studies. In March 2008, researchers took skin cells from mice with Parkinson's disease and used them to test stem cell treatments. They inserted the DNA from these skin cells into enucleated eggs—eggs without their nuclei—and successfully created cloned embryos of mice via SCNT. [source: ScienceDaily]
Researchers have successfully created autologous dopamine neurons from stem cells extracted from cloned embryos, targeting the nerve cells damaged in Parkinson's disease. When these new neurons were implanted into mice, the animals showed significant signs of recovery [source: ScienceDaily].
Xenotransplantation, the process of transplanting animal organs into humans, has been explored as a possible solution for organ shortages. But considering that human bodies sometimes reject organs from other humans, one might wonder how they would respond to organs from animals.
In 2002, scientists at the University of Missouri succeeded in cloning pigs that lack one of two genes known as GATA1, which are primarily responsible for triggering rejection responses in humans [source: CNN]. Although primates are genetically closer to humans for xenotransplantation, pigs are considered the next best alternative until monkey cloning becomes a feasible option [source: Human Genome Project].
Future advancements in stem cell therapy for organ regeneration might not even require cloning. In February 2008, researchers at the University of California, Los Angeles discovered how to derive stem cells from adult human skin cells by manipulating four key regulatory genes that control cell differentiation [source: ScienceDaily].
By reprogramming skin cells to behave like stem cells, these altered cells became pluripotent and were classified as induced pluripotent stem cells. A few months later, Dutch researchers used leftover cellular material from open heart surgeries to extract adult stem cells, which they then used to cultivate heart muscle cells, all without resorting to embryonic stem cells or cloning [source: ScienceDaily].
Due to the ethical concerns surrounding embryonic stem cell research, many people have shown greater support for alternative approaches, such as those mentioned above. In theory, it should eventually be possible to grow new organs from stem cells. However, the technological breakthroughs discussed suggest that cloning may not be necessary to utilize these valuable cells.
