All the things we've been taught about water during our school years seem so straightforward and logical, don’t they? It always seeks its lowest point. It makes up the largest portion of the human body. It covers most of Earth’s surface! It’s crucial for sustaining life, from plants to animals, and everything in between. Even in health class, we’ve known for years that water is vital for weight management, proper organ function, brain health, athletic performance, and much more. But there’s still so much more to discover!
In this list, we’re about to explore ten amazing facts about water that you probably haven’t heard before. These aren’t the basic, surface-level facts you learn about good old H2O in school. Instead, we’re diving deep into more technical and fascinating knowledge surrounding water and its unique properties. Attention, science enthusiasts: we’ve got water on our minds!
10. Hot Water Freezes Faster?!

The Mpemba effect is a curious phenomenon, named after Tanzanian student Erasto B. Mpemba. This effect challenges the common understanding of hot and cold water. You’d naturally assume that cold water freezes faster than hot water, wouldn’t you? Well, it turns out that the opposite is true!
It’s generally believed that cold water freezes faster than hot water, right? Surprisingly, hot water can sometimes freeze faster than cold water. Though this concept seems paradoxical, it’s been documented for centuries—and now, it’s named after Mpemba, a student who helped catalogue the phenomenon decades ago.
There are several factors that contribute to the Mpemba effect, such as differences in evaporation rates, the formation of ice crystals, and the cooling rate. However, the exact conditions under which hot water freezes faster than cold water can vary, which is why this phenomenon remains an intricate puzzle for scientists to solve.
In fact, some scientists still debate whether the Mpemba effect is real and reliably reproducible! Nevertheless, understanding this effect could lead to practical applications, such as improving ice-making processes and optimizing cooling methods in industries related to food production and manufacturing.
9. Water Isn’t Perfect

When we think of pure water, we often picture it as simply H2O. The purest form! That’s how bottled water commercials often portray it, right? But in reality, water molecules can vary slightly due to the presence of different isotopes.
Take heavy water, for instance. Scientifically known as deuterium oxide (D2O), it substitutes hydrogen atoms with deuterium, a hydrogen isotope containing one proton and one neutron. Due to the larger mass of deuterium, heavy water is denser than regular water. While the difference may seem subtle, it plays a significant role in nuclear reactions and certain chemical processes.
Heavy water is used in nuclear reactors and as a neutron moderator. It’s still water, maintaining all the properties of regular water. But you definitely wouldn’t want to drink it! The presence of deuterium in heavy water gives it unique properties, which further exemplifies the intricate nature of even the simplest substances in the natural world.
8. Water with Negative Temperatures?!

Water can challenge conventional temperature norms under extreme conditions. Here’s a fascinating fact: water can exist in a negative temperature state under certain conditions. It’s possible to cool water below 32°F (0°C) without it turning into ice.
This phenomenon is known as “supercooling.” At standard pressure, under the right conditions, water can be “supercooled” to temperatures lower than -40°F (-40°C) while still retaining the properties of a liquid.
Researchers have discovered that water can remain in liquid form as low as -55°F (-48°C) before it inevitably freezes. But how does this occur? One key factor is that water typically needs a surface to freeze onto. Water molecules need something solid to attach to in order to start the freezing process, causing them to spread out as they change into ice.
In extremely pure water, free from dust, dirt, or other impurities that could provide a surface for the water molecules to adhere to, the liquid can remain unfrozen even at temperatures far below the normal freezing point.
7. Superionic Ice

In the harsh conditions of giant gas planets like Uranus and Neptune, a rare and fascinating type of ice called superionic ice is found. This unique material possesses characteristics of both solids and liquids, which profoundly challenges our conventional understanding of matter.
Superionic ice forms under extreme temperatures and pressures, with water molecules arranged in a crystalline structure. However, within this lattice, the hydrogen ions behave like a liquid, moving freely between the oxygen ions. Imagine a solid lattice of oxygen, as in normal water, but suspended in a sea of hydrogen ions that exhibit behavior unlike what we typically associate with solid or liquid states.
Scientists are still working to unravel the full implications of this discovery for the universe. While they are fairly confident it’s occurring on Uranus and Neptune, the fact remains that we can’t exactly travel there to set up camp and verify the findings. Nevertheless, studying superionic ice offers valuable insights into the extreme conditions of the outer solar system. It also underscores the incredible adaptability of water in scenarios far beyond the conditions we experience here on Earth!
6. Yes, Water Can Catch Fire
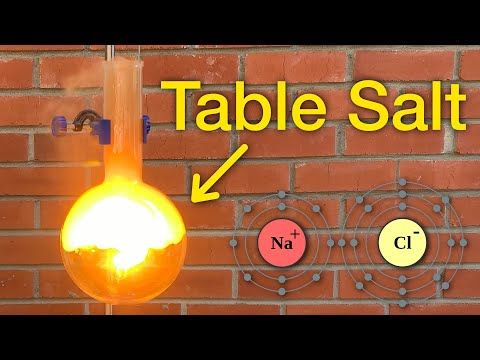
Water is typically known for its ability to extinguish fires, but under specific circumstances, it can actually become involved in a fiery reaction. Sodium, a highly reactive alkali metal, can react explosively when it comes into contact with water. This interaction generates hydrogen gas and heat, leading to fiery explosions.
This counterintuitive phenomenon occurs because sodium ions (Na+) replace the hydrogen ions (H+) in water. This displacement produces hydrogen gas (H2) and generates significant heat. When hydrogen combines with oxygen in the air, it can ignite combustion, creating the ideal conditions for a violent sodium fire.
Handling reactive substances like sodium with great care is crucial, as the interaction with water can be incredibly dangerous. This serves as a potent reminder of the complexity of chemical reactions and the need for stringent safety measures when dealing with reactive materials. It also teaches us that appearances can be deceiving: even water, which we often associate with putting out fires, can become dangerously flammable under the right (or wrong) conditions!
5. The 'Fifth State' of Matter

Most people are familiar with the four classic states of matter: solids, liquids, gases, and plasmas. But within the world of quantum physics, there exists a theoretical 'fifth state' of matter known as Bose-Einstein condensate (BEC). BEC occurs under extremely low temperatures, close to absolute zero, where atoms lose their individual identities and merge into a singular quantum entity. In this state, matter behaves more like a wave than as separate particles, giving rise to intriguing phenomena like superfluidity and quantum interference.
This unique state of matter has become a hot topic of research, with potential applications in fields such as atomic physics and the development of ultra-sensitive measurement devices. Water is often used in experiments, as it is readily available and can be cooled to near absolute zero by scientists.
Since BECs were first discovered only about 30 years ago, there remains much for scientists to explore regarding this 'fifth state' and its implications for future technological innovations.
4. The Universal Solvent

Water is often called the 'universal solvent' because it can dissolve more substances than any other liquid. This remarkable ability comes from its polar nature, with the oxygen atom carrying a slight negative charge and the hydrogen atoms a slight positive charge. This gives water the unique capability to dissolve a wide variety of substances, making it the most effective solvent on Earth. It's not just a title—water literally dominates this category.
Thanks to its polarity, water can interact with other polar or ionic compounds, breaking them down into individual ions and molecules. This feature makes water vital for countless chemical reactions in living organisms, including those that occur in the human body. It's also essential in processes like digestion and cellular activities.
Water’s ability to dissolve an impressive range of compounds—ranging from salts and sugars to acids and bases—highlights its versatility and significance in both nature and industry. This is exactly why staying hydrated is so important! Within your digestive system, water's solvent properties help your body operate efficiently and maintain overall health.
3. The Mystery of Water’s Memory

Here's a fascinating and somewhat controversial concept: 'Water Memory.' This theory suggests that water can 'remember' information and energy. The idea gained attention through the work of Dr. Masaru Emoto, a Japanese researcher and author, who proposed that water could form different crystalline structures when exposed to various stimuli, such as words, music, and thoughts.
Although the concept of water memory is met with skepticism in scientific circles, certain experiments suggest that water may indeed be able to respond to external influences. The hydrogen bonds in water's molecular structure could be responsible for its capacity to record information. If proven true, water memory could have far-reaching effects, potentially influencing fields such as homeopathy and alternative medicine.
Dr. Emoto's research on water memory may not have widespread acceptance in the scientific community, but it has gained enough attention to become an intriguing area of study. Could water 'know' how to react to various external stimuli? Could it 'recognize' these factors in the same way humans do? The science says... maybe!
2. Water's Triple Point

Water possesses a fascinating property known as the 'triple point.' This occurs at exactly 32.018°F (0.01°C) and 611.657 pascals (0.00604 atmospheres) of pressure. At this precise combination of temperature and pressure, water can exist in all three states—solid, liquid, and gas—simultaneously.
This unique phenomenon allows scientists to define the core temperature and pressure values for the solid, liquid, and gas phases of water. The triple point of water is a critical reference for calibrating temperature and pressure measurement instruments used in industries such as manufacturing and mechanical engineering, as well as in the International Temperature Scale (ITS-90).
In practical terms, the triple point of water plays a crucial role in the creation and calibration of thermometers and barometers. For example, the Celsius scale, which is based on the triple point of water (0.01°C), provides a reliable and universally accepted system for measuring temperature. To measure temperature accurately, we need to start from a fixed point, and water’s remarkable properties provide just that starting point.
The triple point of water holds more significance than just for metrology (the science of measurement). It is a fundamental principle in understanding phase transitions and the behavior of matter under different conditions. It reveals the delicate balance between temperature and pressure that determines whether water will exist as a solid, liquid, or gas.
1. Sea Ice Is Freshwater

Although the vast ocean is filled with salty seawater, sea ice itself is made of freshwater. When seawater freezes, a phenomenon known as freezing-point depression occurs. This is because the dissolved salts in seawater lower its freezing point. As the seawater begins to freeze, the ice that forms consists of pure freshwater.
The salts are excluded from the ice crystals, remaining in the remaining liquid ocean water. As a result, the water that surrounds the ice becomes saltier and denser. The sea ice that has formed is almost entirely freshwater, with no salt in its structure.
The reason sea ice is such an important source of freshwater in polar regions is due to the separation of salt from the ice. As you may have guessed (and you're absolutely right), this sea ice can be melted and turned into drinking water and used for other essential needs like cooking and cleaning.
This little fact about sea ice has been a lifesaver for countless generations living in the far northern and southern parts of the Earth. Grasping the science behind sea ice and its freshwater properties is essential for the people in the Arctic and Antarctic, where freshwater resources are incredibly scarce.
