 Diatomic elements are molecules that always consist of exactly two atoms. This rule never changes. - Mytour
Diatomic elements are molecules that always consist of exactly two atoms. This rule never changes. - MytourDiatomic elements detest solitude — they’re never found as single atoms.
Instead, they always appear as pairs of atoms from the same pure element, bonded together. The name says it all: Di means "two," and atomic refers to "atoms." Elements are the fundamental building blocks of the universe.
What Are the 7 Diatomic Elements?
On the periodic table, which consists of 118 elements, only seven of them are diatomic elements:
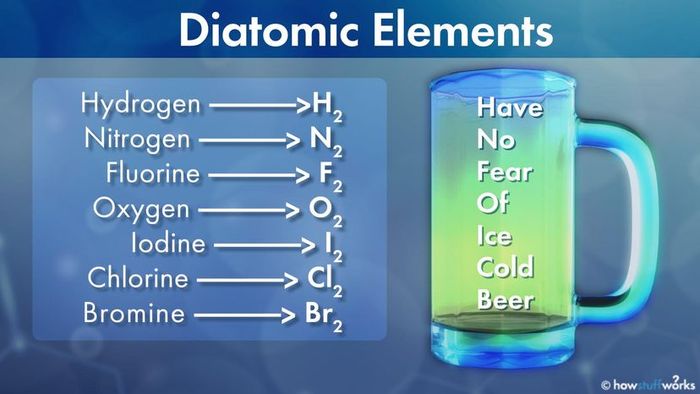
- Hydrogen: H2
- Nitrogen: N2
- Fluorine: F2
- Oxygen: O2
- Iodine: I2
- Chlorine: Cl2
- Bromine: Br2
Aside from hydrogen, which is located at the top-left of the periodic table, the other diatomic elements form a shape resembling an inverted L. So, spotting one will lead you to the rest.
How to Remember the 7 Diatomic Elements
Need an easy way to remember these seven? Use this mnemonic: Have No Fear Of Ice Cold Beer. The first letter of each word will trigger the memory of each diatomic element.
Diatomic Elements Are Everywhere
There are just seven diatomic elements that naturally form this double bond. Of these, five — hydrogen, nitrogen, fluorine, oxygen, and chlorine — exist as gases at standard temperature and pressure. They’re often called "elemental gases."
Bromine is always in liquid form, while iodine can be either liquid or solid at room temperature, depending on various conditions. All seven elements are nonmetals.
Diatomic elements aren’t rare — quite the opposite! Nitrogen and oxygen, in their diatomic forms N2 and O2, make up 99 percent of the Earth's atmosphere. That’s far from being rare.
Diatomic Molecules
Other elements can combine with diatomic elements to create diatomic molecules. This is how we get table salt (sodium + chlorine = NaCl, sodium chloride).
Diatomic molecules are everywhere. While other elements can form diatomic molecules, the bonds are usually weak and unstable, causing them to break apart quickly. Only the seven diatomic elements create strong, lasting bonds and exist in this form almost all the time.
Elements can also be monatomic, meaning they consist of a single atom. (Mon means "one.") Helium is a monatomic element. Additionally, oxygen can be triatomic, with the oxygen atoms creating a triple bond, which we commonly call "ozone."
