 Bohr's Atomic Model. Image Courtesy of Wikimedia Commons (CC BY 4.0)
Bohr's Atomic Model. Image Courtesy of Wikimedia Commons (CC BY 4.0)Essential Insights
- Niels Bohr's atomic model, which showed electrons moving around the nucleus like planets orbiting the sun, earned him the Nobel Prize in 1922, even though it was later proven to be flawed.
- In 1916, Arnold Sommerfeld built upon Bohr's work, introducing elliptical orbits for the electrons.
- Despite its flaws, the Bohr model continues to be a crucial educational tool for explaining atomic structure.
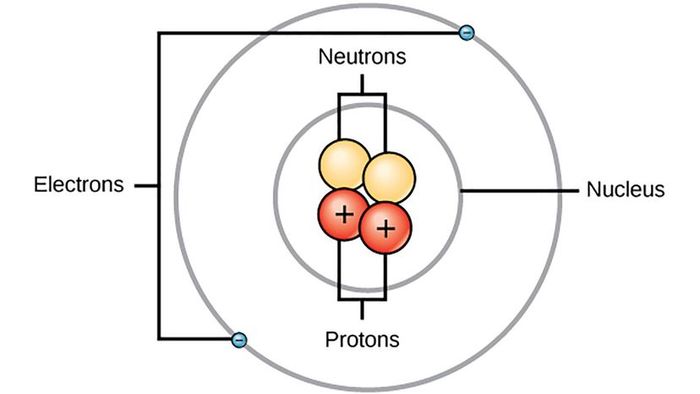
If you search for an image of an atom on the internet, you'll come across one, even though no one has ever directly observed an atom. The idea of what a single atom looks like is based on estimations made by scientists like Danish physicist Niels Bohr.
Atoms are the fundamental building blocks of matter. A single atom of any element is the smallest unit that still follows the laws of physics observable in everyday life (subatomic particles within atoms follow their own unique set of rules). For centuries, scientists speculated about atoms' existence before understanding their structure. Even the ancient Greeks believed that the universe was made up of indivisible components, calling them atomos, meaning "undivided." By the late 1800s, it became clear that chemical substances could be broken down into atoms, which were extremely tiny, and that atoms of different elements had distinct and measurable weights.
In 1897, British physicist J.J. Thomson discovered electrons, negatively charged particles inside atoms. Prior to this, scientists thought atoms were indivisible. While Thomson proposed the existence of electrons, he couldn’t quite determine how they fit into the atom. His best guess was the 'plum pudding model,' imagining the atom as a positively charged sphere with negative particles distributed throughout like fruit in a traditional dessert.
"Electrons are negatively charged and are very small in comparison to atoms," says Dudley Herschbach, a Harvard chemist who won the Nobel Prize in Chemistry in 1986 for his work on the dynamics of chemical elementary processes. "In 1911, Ernest Rutherford discovered the nucleus, which was positively charged. Nuclei have various masses but are significantly larger than electrons, yet still very small in size."
A Giant Leap Forward
In 1912, Niels Bohr, a former student of Rutherford, courageously took over his mentor’s research on unraveling the atom’s structure. Within just a year, he developed a functional model of the hydrogen atom.
 Niels Henrik Bohr (1885-1962), a physicist from Denmark, is renowned for his atomic model and for winning the Nobel Prize in Physics in 1922.
Photo 12/Getty Images
Niels Henrik Bohr (1885-1962), a physicist from Denmark, is renowned for his atomic model and for winning the Nobel Prize in Physics in 1922.
Photo 12/Getty Images"Bohr's 1913 model for the hydrogen atom depicted electrons in circular orbits around the proton, similar to how Earth orbits the sun," explains Herschbach. "He based this on a clear and regular pattern in the hydrogen atom's spectrum, discovered by Johann Balmer in 1885. Additionally, Bohr incorporated Max Planck’s quantum theory from 1900."
Bohr's 1913 model marked a monumental step in atomic theory by integrating the emerging field of quantum mechanics into the description of atoms and molecules. That same year, he published three papers on the structure of atoms and molecules. The most prominent of these focused on the hydrogen atom, while the other two extended his model to elements with additional electrons. His hydrogen atom model proposed electrons moving around the nucleus on specific energy levels, and light was emitted when an electron transitioned from a higher to a lower energy level — a phenomenon that caused hydrogen to glow in a glass tube. Although the model was accurate for hydrogen, it had some flaws.
"Bohr's model struggled to accurately predict the ground-state energies of many-electron atoms and the binding energies of molecules — even with simple two-electron systems like the helium atom or a hydrogen molecule," says Anatoly Svidzinsky, a professor in the Institute for Quantum Science and Engineering at Texas A&M, in an email interview. "By 1913, it was evident that Bohr's model was flawed. For example, Bohr's model wrongly suggests that the hydrogen atom's ground state has nonzero orbital angular momentum."
The 1922 Nobel Prize
This may not make much sense to you if you're not a quantum physicist. But, Bohr's model was awarded the Nobel Prize in Physics in 1922. However, while Bohr solidified his standing in the world of physics, other scientists were refining his model:
"Bohr's model of the hydrogen atom was enhanced by Arnold Sommerfeld in 1916," says Herschbach. "He proposed elliptical orbits, which accounted for spectral lines that were close to those derived from circular orbits. The Bohr-Sommerfeld model of the hydrogen atom is fundamental, though quantum mechanics and relativity later became crucial components."
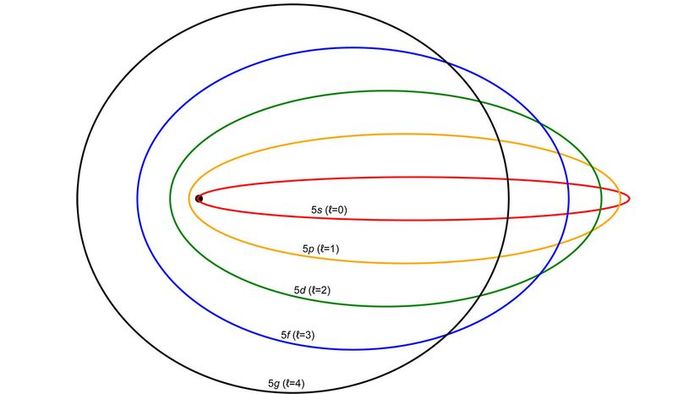 The Sommerfeld model, which refined the Bohr model in 1916 by introducing semi-classical electron orbits.
The Sommerfeld model, which refined the Bohr model in 1916 by introducing semi-classical electron orbits.Between 1925 and 1928, Werner Heisenberg, Max Born, Wolfgang Pauli, Erwin Schrodinger, and Paul Dirac advanced these concepts far beyond Bohr's atomic model, but Bohr's version remains the most well-known model of the atom. The atomic models developed by quantum physics now resemble modern art more than the traditional depiction of a sun surrounded by orbiting electrons. It's likely that the Bohr model persists because it's an effective introduction to atomic theory.
"In 1913, Bohr's model proved that quantization was the correct approach for describing the micro-world," says Svidzinsky. "Thus, Bohr's model guided scientists toward a new direction and encouraged the further development of quantum mechanics. Once the path is known, the correct solution will eventually be discovered. You could think of Bohr's model as a signpost along a hiking trail into the quantum world."
Niels Bohr's father, Christian Bohr, was nominated for three separate Nobel Prizes in Physiology or Medicine, but never won.
