 The AbioCor marked a significant milestone as the first artificial heart utilized in almost 20 years. Explore more images of modern medical advancements.
Photo courtesy Abiomed
The AbioCor marked a significant milestone as the first artificial heart utilized in almost 20 years. Explore more images of modern medical advancements.
Photo courtesy AbiomedEssential Insights
- The AbioCor artificial heart has the potential to extend the life expectancy of end-stage heart failure patients by up to twice as long.
- To qualify for the AbioCor artificial heart, patients must have end-stage heart failure, a prognosis of fewer than 30 days to live, and no alternative treatment possibilities.
- Robert Tools was the first individual to receive the AbioCor artificial heart. Despite facing infections and requiring a ventilator after surgery, his mechanical heart operated flawlessly.
Your heart acts as the body's internal engine, ensuring all systems function smoothly. Essentially, this muscular pump circulates oxygen and blood throughout your lungs and body. Daily, it moves approximately 2,000 gallons of blood. Similar to any engine, neglecting heart health can lead to inefficiencies and breakdowns, a condition known as heart failure.
For many years, heart transplantation was the sole solution for patients suffering from severe heart failure. Yet, with only a little over 2,000 heart transplants conducted each year in the U.S., countless individuals succumb to the wait for a donor heart. A breakthrough occurred on July 2, 2001, when surgeons at Jewish Hospital in Louisville, Kentucky, executed the first artificial heart transplant in almost 20 years. The AbioCor Implantable Replacement Heart, being the first fully autonomous artificial heart, is anticipated to significantly extend the lives of heart patients, potentially doubling their life expectancy.
This article delves into the mechanics of the artificial heart, detailing its functionality, the surgical procedure for chest implantation, and the criteria for potential recipients. Additionally, it contrasts the AbioCor heart with earlier unsuccessful attempts at artificial heart technology.
A Heart Powered by Hydraulics
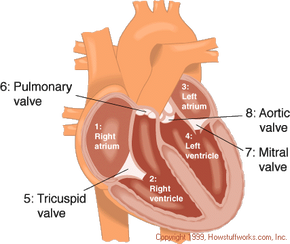 Compartments of the Human Heart
Compartments of the Human HeartAn adult heart typically beats at a pace of 60 to 100 times per minute. For those familiar with How Your Heart Works, it's understood that the heart's contraction occurs in two distinct phases:
- During the initial phase, both the right and left atria simultaneously contract, pushing blood into the right and left ventricles.
- In the subsequent phase, the ventricles contract in unison to eject blood from the heart.
Following this, the heart muscle relaxes in preparation for the next beat, allowing blood to refill the heart chambers.
Individuals with an AbioCor heart implant retain functioning atria that contract simultaneously. However, the artificial heart, which substitutes both ventricles, can only expel blood from one ventricle at a time. Consequently, it sequentially directs blood to the lungs and then to the body, unlike a natural heart that performs both actions simultaneously. The AbioCor is capable of pumping over 10 liters per minute, sufficient for routine activities.
 Illustration of the AbioCor Heart
Source: AbioCor
Illustration of the AbioCor Heart
Source: AbioCorThe AbioCor, crafted by Abiomed, represents a pinnacle of medical engineering, with its central operation relying on a hydraulic pump that moves fluid back and forth. To grasp its functionality, let's examine the system's various components:
- Hydraulic pump - This mechanism operates similarly to hydraulic pumps in heavy machinery, where force applied at one location is transferred to another using an incompressible fluid. A gear within the pump rotates at 10,000 revolutions per minute (rpm) to generate pressure.
- Porting valve - This valve regulates the flow of hydraulic fluid between the two sides of the artificial heart. When fluid shifts to the right, blood is directed to the lungs via an artificial ventricle. Conversely, when fluid moves to the left, blood is pumped to the body.
- Wireless energy-transfer system - Known as the Transcutaneous Energy Transfer (TET), this setup includes two coils (one internal and one external) that use magnetic force to transmit power from an external battery through the skin without breaking it. The internal coil receives this power and channels it to the internal battery and controller.
- Internal battery - A rechargeable battery is placed inside the patient's abdomen, providing 30 to 40 minutes of power for activities like showering when disconnected from the main battery pack.
- External battery - Worn on a Velcro belt around the waist, this rechargeable battery supplies approximately four to five hours of energy.
- Controller - Implanted in the abdominal wall, this compact electronic device oversees and adjusts the heart's pumping rate.
The AbioCor heart, made from titanium and plastic, is linked to four key areas:
- Right atrium
- Left atrium
- Aorta
- Pulmonary artery
The entire system has a weight of around 2 pounds (0.9 kilograms). In the following section, you'll discover how surgeons implanted the AbioCor heart during a seven-hour procedure.
The Seven-hour Surgical Procedure
 Surgeons performing the AbioCor heart implantation
Photo courtesy Abiomed
Surgeons performing the AbioCor heart implantation
Photo courtesy AbiomedThe procedure to implant the AbioCor artificial heart is highly intricate. Surgeons must remove the natural heart's right and left ventricles and replace them with the artificial device. The patient is connected to, and later disconnected from, a heart-lung machine. Hundreds of stitches are required to secure the heart to the artificial ventricles, and grafts—synthetic tissues—are used to link the AbioCor to the remaining parts of the natural heart.
Given the surgery's complexity, a large team of medical professionals is involved. The groundbreaking procedure on July 2, 2001, involved two lead surgeons, 14 nurses, perfusionists, anesthesiologists, and additional support staff.
The steps of the procedure, as outlined by University of Louisville surgeon Robert Dowling, are as follows:
- The energy-transfer coil is implanted in the abdomen.
- The breastbone is opened, and the patient is connected to a heart-lung machine.
- The right and left ventricles of the natural heart are removed, while the atria, aorta, and pulmonary artery remain. This phase alone takes two to three hours.
- Atrial cuffs are attached to the natural heart's right and left atria.
- A plastic model is used to determine the optimal placement of the artificial heart.
- Grafts are trimmed and sewn to the aorta and pulmonary artery.
- The AbioCor is positioned in the chest and connected to the pulmonary artery, aorta, and atria using "quick connects."
- Air is removed from the device.
- The patient is disconnected from the heart-lung machine.
- The surgical team verifies the heart's proper functioning.
Following the initial transplant, additional procedures have been successfully performed.
Initially, Abiomed officials advised against overly hopeful outcomes, with the most optimistic estimates suggesting patients might survive up to six months with the AbioCor heart. The device aims to double the life expectancy of patients who had only about 30 days to live prior to the surgery.
Robert Tools, the first recipient of the AbioCor heart on July 2, 2001, at Jewish Hospital in Louisville, has since passed away. Ten other patients have also died, but on average, recipients of the AbioCor heart lived for five months post-transplant.
In 1982, Dr. William Devries implanted the Jarvik-7, the first device intended as a permanent heart replacement. The surgery took place at the University of Utah, with Dr. Barney Clark, a Seattle dentist, as the patient. Clark survived for 112 days before complications from the device led to his death.
The Jarvik-7, an air-powered heart developed by Dr. Willem Kolff and Dr. Don Olsen, required multiple external wires connected to a large unit outside the body. These wires caused several infections in Clark.
After four more patients received the Jarvik-7, it was discontinued due to issues like stroke, mechanical failure, and fit problems. It has since been improved and renamed the CardioWest heart, now used only in experimental settings and as a bridge-to-transplant device.
The Most Critically Ill
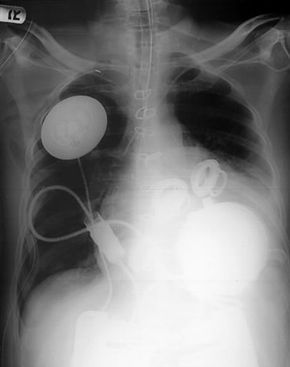 X-ray image showing the AbioCor device implanted in a patient
Photo courtesy Abiomed
X-ray image showing the AbioCor device implanted in a patient
Photo courtesy AbiomedThe U.S. Food and Drug Administration (FDA) has authorized Abiomed to conduct 15 implants as part of a clinical trial. Twelve of these surgeries have already been completed at medical centers in Houston, Los Angeles, Boston, and Philadelphia. The FDA is evaluating the outcomes of these procedures individually to decide the future of the AbioCor device. If the device, costing between $70,000 and $100,000, proves successful in extending patients' lives without complications, it could gain approval for broader use across the U.S. Reports suggest Abiomed plans to seek FDA approval this year to market the AbioCor heart under a "Humanitarian Device Exemption," targeting patients with end-stage heart failure.
Laman Gray stated that the initial candidates for the artificial heart would be the "most critically ill." The FDA and Abiomed have established specific criteria for selecting recipients. Patients must meet the following conditions:
- Suffer from end-stage heart failure
- Have a life expectancy of fewer than 30 days
- Be ineligible for a natural heart transplant
- Have no other viable treatment options
Additionally, the grapefruit-sized device must fit within the patient's chest. To assess compatibility, patients undergo a CAT scan and chest X-ray. Using a computer-aided design (CAD) program, doctors simulate the removal of the natural heart and the placement of the AbioCor heart. If the simulation confirms a proper fit, the surgical team can proceed with the implantation.
For almost two months after the procedure, the identity of the first recipient remained confidential. However, on August 21, 2001, it was disclosed that Robert Tools, a Kentucky native and former telephone company worker, was the groundbreaking patient. Despite battling an infection and requiring a ventilator post-surgery, his doctors confirmed that the mechanical heart was functioning flawlessly.
 Patient Robert Tools alongside Dr. Laman Gray (left) and Dr. Robert Dowling (right)
Photo courtesy Jewish Hospital
Patient Robert Tools alongside Dr. Laman Gray (left) and Dr. Robert Dowling (right)
Photo courtesy Jewish HospitalHere are the details known about Tools before his surgery:
- He was diagnosed with class IV heart failure.
- He experienced severe bi-ventricular failure.
- He was rejected by a heart transplant center.
- He had undergone prior coronary bypass surgery.
- He had suffered multiple heart attacks.
- He had diabetes.
Currently, between 2 million and 3 million Americans are living with heart failure, with 400,000 new cases diagnosed each year. According to the National Heart, Lung, and Blood Institute (NHLBI), heart failure is responsible for 39,000 deaths annually. Most individuals diagnosed with heart failure have a life expectancy of about five years and typically require a heart transplant to prolong their survival.
In 2003, 2,143 heart transplants were conducted in the United States. Despite this, thousands of individuals die each year while awaiting a donor heart. While doctors continue to advocate for organ donation, the AbioCor offers hope to those who cannot receive a natural transplant or wait for a suitable donor heart.
