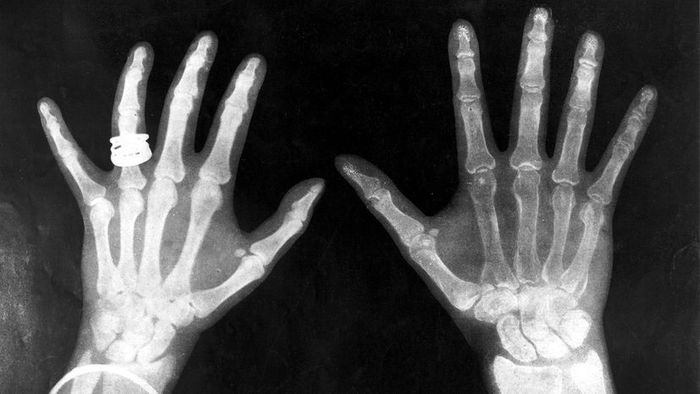
 This X-ray, depicting the hands of Queen Mary and King George, was captured just a year after the accidental yet groundbreaking discovery of X-rays in 1895. Science & Society Picture Library/SSPL via Getty Images
This X-ray, depicting the hands of Queen Mary and King George, was captured just a year after the accidental yet groundbreaking discovery of X-rays in 1895. Science & Society Picture Library/SSPL via Getty ImagesLike many great discoveries in history, X-ray technology was invented by sheer accident. In 1895, German physicist Wilhelm Roentgen stumbled upon X-rays while working with electron beams inside a gas discharge tube. He observed a fluorescent screen in his lab glowing when the electron beam was activated. While this wasn’t unexpected—since fluorescent materials typically light up when exposed to electromagnetic radiation—Roentgen's tube was surrounded by thick black cardboard, which he assumed would block most of the radiation.
Roentgen then experimented by placing various objects between the tube and the screen, and the screen continued to glow. Eventually, he positioned his hand in front of the tube and saw the shadow of his bones projected on the screen. In that moment, he not only discovered X-rays but also uncovered their most valuable application.
Roentgen's extraordinary discovery paved the way for one of the most significant breakthroughs in medical history. X-ray technology allows doctors to peer through human tissue, making it possible to examine broken bones, locate cavities, and identify swallowed objects with remarkable precision. With specialized X-ray techniques, it is also possible to investigate softer tissues such as the lungs, blood vessels, and intestines.
In this article, we will explore the simple yet fascinating process behind how X-ray machines perform this astonishing feat. It turns out, the fundamental mechanism is surprisingly straightforward.
What exactly is an X-ray?
X-rays are fundamentally similar to visible light rays. Both are types of electromagnetic energy that travel in waves and are carried by particles called photons (see How Light Works for more details). The key difference between X-rays and visible light lies in the energy level of the photons, which is also reflected in the rays' wavelength.
Our eyes are tuned to detect the specific wavelength of visible light, but we cannot perceive the shorter wavelength of higher-energy X-rays or the longer wavelength of lower-energy radio waves.
Both visible light photons and X-ray photons are produced by the movement of electrons within atoms. Electrons exist in different energy levels or orbitals around an atom's nucleus. When an electron moves to a lower orbital, it releases the excess energy in the form of a photon. The energy of the photon depends on how far the electron has moved between orbitals. (For more details on this process, refer to this page.)
When a photon strikes an atom, the atom may absorb the photon's energy, causing an electron to jump to a higher energy level. For this to happen, the energy of the photon must match the energy difference between the two electron positions. If the energy levels don't align, the photon cannot move the electrons between orbitals.
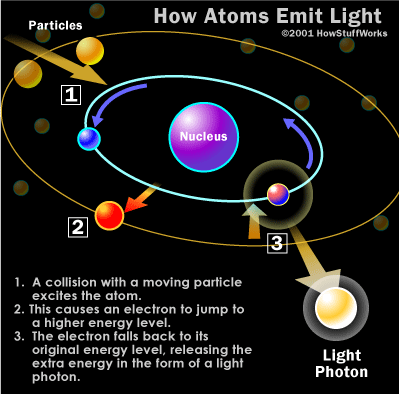
The atoms that make up your body tissue are efficient at absorbing visible light photons. The energy of these photons corresponds well with the energy differences between electron positions. Radio waves, on the other hand, lack the energy to shift electrons between orbitals in larger atoms, so they pass through most materials. X-ray photons, though, are able to pass through most things for the opposite reason: their energy is too high.
However, X-ray photons can knock electrons out of atoms entirely. Some of the photon's energy is used to eject an electron from the atom, while the rest propels the electron through space. Larger atoms are more likely to absorb X-ray photons in this way, as they have greater energy differences between their orbitals, which better matches the energy of the photon. Smaller atoms, with their smaller energy gaps between orbitals, are less likely to absorb X-ray photons.
Soft tissues in your body consist of smaller atoms, which don't absorb X-ray photons very effectively. On the other hand, the calcium atoms in your bones are much larger, making them much better at absorbing X-ray photons.
In the following section, we'll explore how X-ray machines apply this effect in practice.
While the most significant impact of X-ray technology has been in the medical field, X-rays have also played a vital role in several other domains. They have been instrumental in research related to quantum mechanics, crystallography, and cosmology. In industry, X-ray scanners are commonly used to identify tiny defects in heavy machinery. Additionally, X-ray scanners are now a staple in airport security systems.
How the X-Ray Machine Works
At the core of an X-ray machine is a pair of electrodes – a cathode and an anode – housed within a glass vacuum tube. The cathode is a heated filament, much like the one found in older fluorescent lamps. A current passes through the filament, causing it to heat up, which in turn causes the filament to emit electrons. The positively charged anode, a flat disc made of tungsten, draws the electrons across the tube.
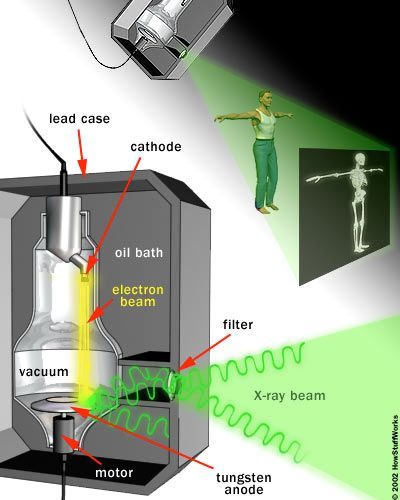 X-ray machines seem to perform the impossible: They can peer through clothing, flesh, and even metal, thanks to some fascinating scientific principles. Discover how these machines are able to reveal the bones beneath your skin.
X-ray machines seem to perform the impossible: They can peer through clothing, flesh, and even metal, thanks to some fascinating scientific principles. Discover how these machines are able to reveal the bones beneath your skin.The voltage difference between the cathode and anode is incredibly high, causing the electrons to accelerate rapidly through the tube. When a fast-moving electron collides with a tungsten atom, it dislodges an electron from one of the atom's lower orbitals. An electron from a higher orbital quickly falls to the lower energy level, releasing its excess energy as a photon. Since the drop is substantial, the photon emitted is an X-ray photon with a high energy level.
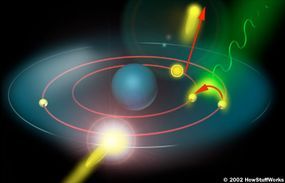
Free electrons can also produce photons without directly striking an atom. The nucleus of an atom may attract a fast-moving electron just enough to alter its path. Similar to how a comet changes direction when passing by the sun, the electron slows down and changes direction as it passes close to the atom. This 'braking' action results in the emission of excess energy in the form of an X-ray photon.
The powerful collisions involved in X-ray production generate significant amounts of heat. To prevent the anode from melting, a motor rotates it (since the electron beam isn't always focused on the same spot). Additionally, a cool oil bath surrounding the tube absorbs the heat.
The entire system is encased in a thick lead shield to prevent the X-rays from radiating in all directions. A small window in the shield allows some X-ray photons to escape in a concentrated beam. This beam then passes through a series of filters before reaching the patient.
A camera positioned on the opposite side of the patient captures the X-ray pattern that passes through the patient's body. The X-ray camera functions similarly to an ordinary camera, but instead of visible light, X-ray photons trigger a chemical reaction on the film. (For more information on this process, see How Photographic Film Works.)
Typically, doctors retain the film image as a negative. This means that areas exposed to more light appear darker, while those exposed to less light appear lighter. Hard materials like bone appear white, while softer tissues show up as black or gray. By adjusting the intensity of the X-ray beam, doctors can bring different materials into sharper focus.
In a standard X-ray, most soft tissues do not show up clearly. To better visualize organs or to study blood vessels in the circulatory system, doctors use contrast media, which are liquids that absorb X-rays more efficiently than surrounding tissues. To highlight organs in the digestive or endocrine systems, a patient typically consumes a contrast media mixture, usually a barium compound. For imaging blood vessels or other parts of the circulatory system, contrast media is injected into the bloodstream. Contrast media are often combined with a fluoroscope. In fluoroscopy, X-rays pass through the body to a fluorescent screen, producing a real-time X-ray image. This method allows doctors to track the flow of contrast media through the body and record the moving X-ray images on film or video.
Are X-Rays Dangerous?
X-rays are an invaluable tool in medicine, allowing doctors to peer inside the body without performing surgery. It's far safer and more convenient to examine a broken bone using X-rays than it is to surgically open up a patient.
However, X-rays can also be harmful. In the early days of X-ray technology, many doctors exposed both patients and themselves to X-ray beams for extended periods. This led to the development of radiation sickness in both doctors and patients, signaling that something was amiss within the practice.
The issue with X-rays lies in the fact that they are a form of ionizing radiation. While regular light doesn’t alter an atom, an X-ray can strike an atom with enough force to knock electrons off, creating an ion—an electrically charged atom. These free electrons can then interact with other atoms, generating additional ions.
The electrical charge of an ion can trigger unnatural chemical reactions within cells. For instance, it can break DNA strands. A cell with a broken DNA strand will either die or develop a mutation. If too many cells die, the body may experience various diseases. If the DNA mutates, the cell could become cancerous, potentially spreading the cancer. A mutation in sperm or egg cells may even result in birth defects. Because of these potential dangers, doctors now use X-rays with caution.
Despite these risks, X-ray scanning remains a safer alternative to surgery. X-ray machines are essential tools in medicine, as well as being valuable in security and scientific research. They truly stand as one of the most beneficial inventions ever created.
For further details on X-rays and X-ray machines, explore the links provided below.
