 Steam billows from the PacifiCorps Hunter coal-powered energy facility near Castle Dale, Utah. Operational since 1978, this 1,577-megawatt plant stands as one of the most significant coal-based energy producers in the western United States. GEORGE FREY/AFP via Getty Images
Steam billows from the PacifiCorps Hunter coal-powered energy facility near Castle Dale, Utah. Operational since 1978, this 1,577-megawatt plant stands as one of the most significant coal-based energy producers in the western United States. GEORGE FREY/AFP via Getty ImagesPicture a world where a villainous mastermind devises a plan to extract all the oxygen from the atmosphere and bury it underground. While this seems like a plot from a comic book when it comes to oxygen, scientists are actively developing methods to achieve something similar with carbon dioxide. The goal? To mitigate global warming and climate change.
Carbon dioxide (CO2) is a naturally occurring gas that permits sunlight to penetrate the Earth's atmosphere while trapping some of the sun's heat, preventing it from escaping back into space. This phenomenon, known as the greenhouse effect, naturally warms the Earth to levels suitable for life. Without it, the planet's average temperature would plummet to 0 degrees Fahrenheit (-18 degrees Celsius) [source: Lang]. While such conditions might be ideal for skiing, life as we know it would cease to exist.
While carbon dioxide and the greenhouse effect are essential for sustaining life on Earth, human activities like burning fossil fuels in power plants and vehicles are releasing excessive amounts of CO2. This overabundance poses a significant threat to our environment.
The period from 2011 to 2020 marked the hottest decade ever recorded [source: World Meteorological Organization]. Since the late 19th century, the Earth's average temperature has increased by approximately 2.12 degrees Fahrenheit (1.18 degrees Celsius) [source: NASA]. This rise has led to polar ice melting, higher sea levels, altered animal migration patterns, and more frequent extreme weather events [sources: Carrington, NOAA, and Bradford].
What is the primary cause of this warming? Human activity. From 1970 to 2004, carbon dioxide emissions surged by 90 percent [source: PBL]. By 2019, the global average CO2 concentration in the atmosphere reached levels higher than any in the past 800,000 years [source: Lindsey].
In recent years, the United Nations' Economic Commission for Europe (UNECE) has advocated for the widespread adoption of carbon capture technology [source: U.N. News].
Carbon capture is a process that involves capturing CO2 at its source, transporting it to a storage site (typically deep underground), and securely isolating it. This method has the potential to prevent excess CO2 from being released into the atmosphere.
This article explores various existing and emerging techniques for capturing and storing carbon.
Trapping Carbon Dioxide: Carbon Capture Technology
 In 2014, the Boundary Dam Power Station near Estevan, Saskatchewan, Canada, became the world's first power plant to implement carbon capture and storage successfully. Generating 115 megawatts of power, it cuts SO2 emissions from coal processing by up to 100 percent and CO2 emissions by up to 90 percent. SaskPower
In 2014, the Boundary Dam Power Station near Estevan, Saskatchewan, Canada, became the world's first power plant to implement carbon capture and storage successfully. Generating 115 megawatts of power, it cuts SO2 emissions from coal processing by up to 100 percent and CO2 emissions by up to 90 percent. SaskPowerCarbon capture and storage (CCS) involves three primary stages:
- capturing and isolating CO2 from other gases
- transporting the captured CO2 to a designated storage site
- storing CO2 away from the atmosphere (underground or deep in the ocean)
Let’s delve deeper into the process of capturing and separating CO2:
Carbon is extracted from power plant emissions through three primary methods: post-combustion, pre-combustion, and oxy-fuel combustion [source: National Energy Technology Laboratory].
A fossil fuel power plant produces energy by burning fossil fuels like coal, oil, or natural gas. This combustion generates heat, which creates steam to drive a turbine connected to an electricity generator. The burning process is also known as combustion.
In post-combustion carbon capture, CO2 is captured after the fossil fuel has been burned. The combustion process produces flue gases, which consist of CO2, water vapor, nitrogen, and sulfur dioxide.
In post-combustion carbon capture, CO2 is isolated and extracted from flue gases produced during fossil fuel combustion. This method is the most widely adopted in carbon capture technology. It’s advantageous because it can be implemented in both new and existing coal-fired power plants. However, it has limitations, such as the need for bulky equipment and reduced turbine efficiency [source: Elhenawy].
Precombustion carbon capture involves capturing and removing carbon from fossil fuels before the combustion process is completed.
Fossil fuels like coal, oil, or natural gas are heated with steam and oxygen, producing a synthesis gas (syngas) composed mainly of CO2, hydrogen (H2), and carbon monoxide (CO). A subsequent reaction converts water (H2O) into hydrogen, while some carbon monoxide is transformed into carbon dioxide. The final mixture is rich in H2 and CO2 [source: U.S. Department of Energy].
The CO2 in this mixture is easily separated, captured, and stored. Meanwhile, the hydrogen can be utilized in other energy production processes.
Precombustion carbon capture is generally more efficient than postcombustion methods. However, it requires costly equipment and is less compatible with older power plants compared to newer facilities [source: Elhenawy].
In oxy-fuel combustion carbon capture, power plants burn fossil fuels in a gas mixture rich in pure oxygen rather than ordinary air. This produces flue gas primarily composed of CO2 and water. The CO2 can then be separated by compressing and cooling the water [sources: National Energy Technology Laboratory and National Resources of Canada].
While some aspects of oxy-fuel combustion carbon capture are cost-effective, the overall process is expensive due to the high cost of pure oxygen. Additionally, its scalability remains uncertain. A 2020 review in the journal Catalysts noted that the technology "needs to be validated for large-scale operations" [source: Elhenawy].
On the upside, oxy-fuel combustion capture is adaptable to both older and newer coal-fired power plants [source: Elhenawy].
A critical question arises: After capturing the carbon, how is it transported to storage sites? Continue reading to discover the answer.
Ground that remains at or below 32 degrees Fahrenheit (0 degrees Celsius) for two consecutive years or more is known as permafrost. This icy terrain has become a focal point in climate change discussions. In the Northern Hemisphere's permafrost regions, approximately 1.6 trillion to 1.7 trillion tons (1,460 billion to 1,600 billion metric tons) of carbon are stored within the soil. As global temperatures rise and these frozen layers thaw, scientists are keen to understand the impact of this released carbon on our planet [source: Schurr].
Transporting Carbon Dioxide
 Pipelines, such as the one at the Boundary Dam Power Station near Estevan, Saskatchewan, Canada, are commonly used to move CO2. Phillip Chin/Shell Canada Limited
Pipelines, such as the one at the Boundary Dam Power Station near Estevan, Saskatchewan, Canada, are commonly used to move CO2. Phillip Chin/Shell Canada LimitedOnce carbon dioxide (CO2) is captured, it must be transported to a storage facility. The most common method for moving CO2 is via pipeline.
Pipelines have been operational for many years, transporting vast quantities of gases, oil, and water daily. CO2 pipelines are already part of the infrastructure in the U.S. and numerous other countries. Currently, there are over 4,039 miles (6,500 kilometers) of CO2 pipelines across Africa, Australia, the Middle East, and North America. Most were built for Enhanced Oil Recovery (EOR), but some are linked to CCS projects [source: Noothout].
Pipelines can be installed almost anywhere, whether underground or underwater. They traverse a variety of landscapes, including deserts, agricultural fields, mountain ranges, and oceans [source: Intergovernmental Panel on Climate Change].
Pipelines are often linked to processing facilities or power plants that use fossil fuels, as well as natural CO2 sources. The purity of the CO2 in the pipeline can vary depending on the technology used at the source [source: Noothout].
Sometimes, CO2 travels as far as possible through a pipeline before being transferred to tanker trucks, ships, or pressurized cylinders for the remainder of its journey. It’s important to note that a large CO2 leak poses an asphyxiation risk. Proper construction and safe handling, similar to transporting natural gas and other hazardous materials, are crucial.
Pipelines can carry CO2 in three forms: gas, liquid, or solid. Solid CO2, also known as dry ice, is not economically viable for transportation.
Most pipelines transport CO2 in its gaseous form, which must be compressed before being moved. The National Energy Technology Laboratory states that the optimal pressure range for transportation is between 1500 and 2200 PSI (or 10,342 and 15,168 KPA).
Engineers must carefully monitor impurities in the CO2 stream, such as hydrogen sulfide and water. Water, in particular, can corrode pipelines and, under high pressure and low temperatures, form natural-gas hydrates—solid crystals that block pipelines. Researchers are actively developing solutions to manage these impurities [sources: Onyebuchi and Bai].
Safety is paramount in pipeline construction. A rupture near populated areas could release large amounts of CO2, posing significant risks to public health and the environment. To prevent accidental damage, pipelines are often buried deep underground, and routes are planned to avoid cities and towns whenever possible [source: Onyebuchi].
DNV, a leading risk management and quality assurance firm in Norway, introduced updated safety protocols for CO2 transport pipelines in 2021. Additionally, the United Kingdom's Health and Safety Executive has comprehensive guidelines addressing issues like corrosion and land use.
The cost of constructing pipelines varies based on factors such as the route (e.g., through urban areas, mountains, or offshore), material quality, equipment used, labor requirements, and other associated expenses.
Carbon atoms make up about 12 percent of the atoms in the human body. For example, a 176-pound (80-kilogram) person contains approximately 31.7 pounds (14.4 kilograms) of carbon. This is a substantial amount, second only to oxygen in terms of body mass. Over 99 percent of the atoms in our bodies are oxygen, carbon, hydrogen, or nitrogen [source: New Scientist].
Carbon Storage
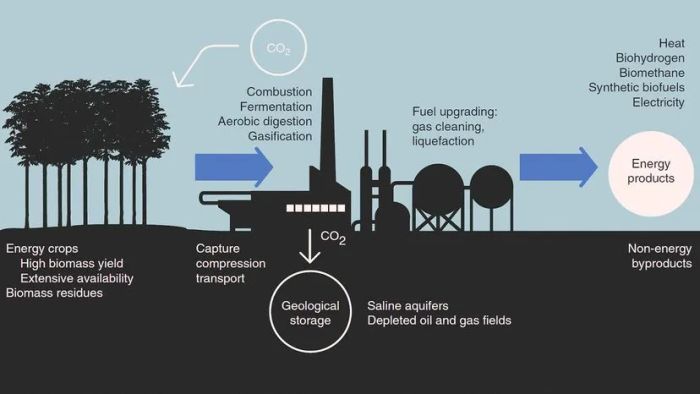 The process of carbon capture and storage explained by The Royal Society of Chemistry
The process of carbon capture and storage explained by The Royal Society of ChemistryOnce carbon dioxide (CO2) is captured and transported, the next challenge is finding a suitable storage location. Will it require massive storage units, desert tanks, or additional landfills for CO2 waste?
Fortunately, the answer is "no." CO2 can be stored in various locations, particularly underground. Research indicates that the United States alone has sufficient subsurface capacity, such as deep aquifers and permeable rocks, to potentially store 1.8 trillion tons (1.71 trillion metric tons) of carbon dioxide [source: Cunliff and Nguyen].
Let’s delve into the details of underground storage. Deep below the surface, CO2 can be stored under pressures exceeding 1,057 PSI (72.9 atm) and temperatures above 88 degrees Fahrenheit (31.1 degrees Celsius).
Under these specific conditions, CO2 transitions into a supercritical state. In this form, it exhibits properties of both gases and liquids. Supercritical CO2 has the low viscosity of a gas but the high density of a liquid [sources: National Energy Technology Laboratory and Imaging Technology Group].
Due to its ability to permeate porous rocks, a significant amount of CO2 can be stored in a compact area. Oil and gas reservoirs are particularly suitable for CO2 storage, as their porous rock layers have historically trapped oil and gas for millennia [source: Center for Science Education].
CO2 is injected into underground rock formations beneath the Earth's surface. These natural reservoirs are sealed by overlying rocks, preventing the gas from escaping. However, there are potential risks associated with underground storage, which we’ll explore later.
Basaltic rock formations are also ideal for CO2 storage. Formed from volcanic activity, basalt is one of the most abundant rock types in the Earth's crust. Research shows that when CO2 reacts with the magnesium and calcium in basalt, it can be converted into solid minerals like dolomite, calcite, and magnesite [source: Cartier].
Another option is coal deposits. Some coal seams deemed "unmineable" can store vast amounts of captured CO2. These deposits allow for storage at lower pressures, reducing costs [source: Talapatra].
Beyond underground storage, the ocean is being explored as a potential site for permanent CO2 storage. One idea involves depositing CO2 deep in the ocean, below 9,842 feet (3,000 meters), where its density exceeds that of water, potentially trapping it for extended periods [source: Center for Science Education].
However, ocean carbon storage remains largely experimental, raising concerns about its impact on marine ecosystems and the risk of CO2 eventually re-entering the atmosphere.
Next, we’ll examine these concerns more closely and assess whether carbon capture and storage is a feasible long-term solution.
Researchers at the University of California, Los Angeles, have proposed a novel method for CO2 removal. This approach involves extracting CO2 from seawater and converting it into limestone and magnesium for storage. Since oceans hold about 150 times more CO2 than the atmosphere, removing CO2 from seawater could encourage the oceans to absorb more atmospheric CO2. Known as "single-step carbon sequestration and storage" (sCS), this process would likely require a massive financial investment, potentially in the trillions of dollars [source: Lewis].
Carbon Storage Concerns
 The Petra Nova facility, a coal-powered plant near Houston, Texas, is the sole carbon capture and storage facility in the U.S. It captures and repurposes over 90 percent of its CO2 emissions. Luke Sharrett/Bloomberg via Getty Images
The Petra Nova facility, a coal-powered plant near Houston, Texas, is the sole carbon capture and storage facility in the U.S. It captures and repurposes over 90 percent of its CO2 emissions. Luke Sharrett/Bloomberg via Getty ImagesWhile carbon capture and storage appears promising, it is not without its challenges and controversies.
First, it’s crucial to understand that carbon capture and storage (CCS) does not justify continued CO2 emissions. Regardless of CCS advancements, other emission-reduction strategies remain essential. That said, CCS offers a way to mitigate emissions from existing power plants.
A 2020 report by the Global CCS Institute reveals that there are currently "65 commercial CCS facilities at various stages of development worldwide."
However, some critics raise concerns about the financial viability of CCS. Unlike electric vehicles and solar panels, which can be marketed and sold to individuals and businesses, monetizing captured CO2 has proven to be a significant challenge.
Another issue is the high energy demand of current CCS technologies. Additionally, these systems rely heavily on water for cooling and processing, which raises further concerns [sources: Magneschi and Rosa].
This reliance on water has sparked debates about whether CCS could exacerbate water scarcity. In 2020, a research team led by Lorenzo Rosa at the University of California, Berkeley, simulated the impact of equipping all large coal-fired power plants globally with four different CCS technologies.
As stated in their paper, published in the journal Nature Sustainability on May 4, 2020, "some regions lack adequate water resources to support the additional water demands of CCS technologies."
This is just one of many environmental concerns surrounding carbon capture and storage.
What if the stored carbon dioxide leaks from underground? Predicting the long-term behavior of CO2 buried beneath the Earth’s surface is challenging. Strong regulations and careful site selection could play a crucial role in preventing future issues.
There are several ways recaptured CO2 might escape to the surface. Ironically, the wells used to inject it underground could become potential leakage points. Abandoned oil and gas wells or natural faults could also serve as escape routes [source: Dunne].
A 2018 study suggests leaks are unlikely if "realistically well-regulated storage" practices are followed. This finding contrasts with some earlier research on the topic [sources: Dunne and Alcalde].
Some critics argue that CCS, whether viable or not, distracts from the real issue. They believe efforts should focus on reducing dependence on fossil fuels, as CCS extends the lifespan of fossil fuel-dependent power plants.
On the opposing side, CCS advocates argue that renewable energy alone isn’t enough. They believe that combining renewables with carbon capture technology is essential to effectively combat catastrophic climate change.
Many uncertainties remain about the ultimate role of carbon capture and storage in mitigating the greenhouse effect and addressing climate change. However, one thing is clear: Carbon dioxide emissions are a global issue.
Trees are valuable allies in the fight against global warming and climate change. Through photosynthesis, they absorb and store carbon dioxide, functioning like natural CCS systems. However, scientists warn that planting trees alone cannot offset the excessive CO2 released by burning fossil fuels. Additionally, older, diverse forests are more effective at sequestering CO2 than younger, less varied ones [source: Tso].
