 If you toss a pot of boiling water into the frigid air during winter, you'll witness a fascinating outcome—assuming it's cold enough. This phenomenon is known as the Mpemba effect. makieni/Shutterstock
If you toss a pot of boiling water into the frigid air during winter, you'll witness a fascinating outcome—assuming it's cold enough. This phenomenon is known as the Mpemba effect. makieni/ShutterstockKey Insights
- The Mpemba effect proposes that hot water may freeze more quickly than cold water under specific conditions, a concept first noted by Aristotle.
- In 1963, Tanzanian student Erasto B. Mpemba rediscovered this effect while making ice cream, marking the first documented instance that sparked scientific exploration.
- Though still a subject of debate, recent studies indicate that the Mpemba effect might occur because hot and cold water reach thermal equilibrium differently.
For centuries, brilliant thinkers from Aristotle to Descartes have entertained the notion that — contrary to conventional wisdom — hot water might actually freeze faster than cold water. Yet, no scientific consensus has ever truly validated this hypothesis.
In 1963, a Tanzanian physics student, Erasto B. Mpemba (pronounced em-pem-ba), reignited this concept after a serendipitous incident while making ice cream at school. His observations seemed to confirm what philosophers like Aristotle and Descartes had once speculated: hot water freezes faster than cold water. Mpemba documented his findings in a 1969 paper, simply titled 'Cool?', which introduced the term 'Mpemba effect.' But, was he right? Does hot water indeed freeze quicker than cold water?
What is the Mpemba Effect?
The Mpemba effect refers to a curious phenomenon in physics where, when hot and cold water are placed in the same freezing environment, the hot water freezes faster than the cold water.
Erasto Mpemba observed that during a school ice cream-making session, when he placed a near-boiling mixture of sugar and milk (mostly water) into a freezer, it froze more quickly than other mixtures that had been cooled to room temperature before freezing.
Mpemba hypothesized from his observations that when equal amounts of water — one at 212 degrees Fahrenheit (100 degrees Celsius) and the other at 95 degrees Fahrenheit (35 degrees Celsius) — were placed in identical beakers and subjected to freezing conditions, the hotter water would freeze faster. His ice cream experiment and the theory about water behavior aligned him with centuries of scientists who had also speculated about this peculiar property of water.
Understanding the Freezing Process
As water transitions into ice, it undergoes a phase change, shifting from a liquid to a solid state. Physicists typically describe the phase of a substance when it reaches equilibrium, indicating that the substance is in a stable state where no significant energy flows from one area to another. At equilibrium, the volume and temperature of the substance remain constant. However, when a substance is out of equilibrium, its energy fluctuates, and so may its state of matter.
For water to freeze and remain frozen, the individual water molecules must reach equilibrium. If excess energy moves through water that is not in equilibrium, it will oscillate between solid and liquid states (at lower temperatures) or liquid and gas states (at higher temperatures). The quicker the water molecules reach equilibrium with lower energy, the faster they can freeze.
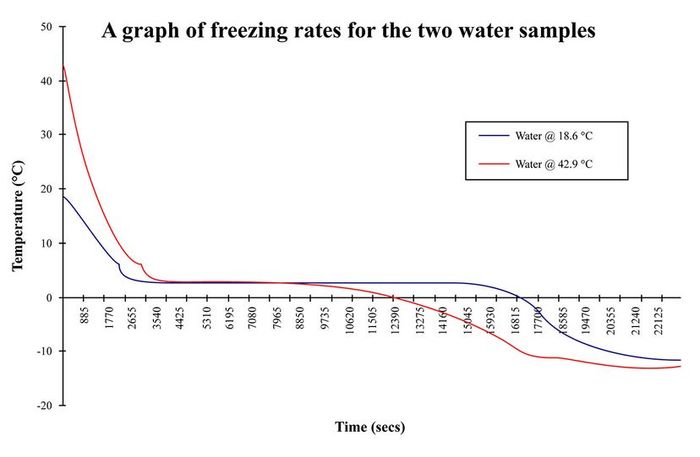 This graph, depicting the freezing rates of two different water samples, illustrates the Mpemba effect.
Wikimedia Commons (CC BY 3.0)
This graph, depicting the freezing rates of two different water samples, illustrates the Mpemba effect.
Wikimedia Commons (CC BY 3.0)How Can Hot Water Freeze Faster Than Cold Water?
Physicists remain divided on whether hot water reliably freezes faster than cold water. When this does occur, specific conditions must be in place for it to happen.
When water is placed in a freezing environment, various sections of the water reach equilibrium at different times. The outer parts of the water cool more rapidly, potentially freezing before the water in the center remains liquid. Moreover, when hot water (such as the 212-degree boiling water mentioned by Mpemba) is placed in a freezer, it also emits steam from the top of the container, reducing the total amount of water that needs to freeze.
Additionally, cold water (or even water at room temperature) often forms a layer of frost on its surface as part of the freezing process. Ironically, this frost acts as a temporary insulator (similar to how an ice igloo protects its inhabitants from cold air), which can slow down the overall freezing. In contrast, hot water, at least during the initial freezing stages, prevents frost formation, allowing cold air to penetrate further into the vessel.
Here are several ways that hot water might freeze faster than cold water. However, it is crucial to remember that for water to freeze and remain frozen, it needs to reach a state of equilibrium.
The Study That May Prove the Mpemba Effect
If there is any evidence supporting the Mpemba effect's reality and consistency, it comes from a 2020 study conducted by John Bechhoefer and Avinash Kumar. Published in the journal Nature, the study involved heating microscopic glass beads in a controlled "energy landscape" influenced by lasers. The researchers varied the beads' temperatures and examined which ones reached equilibrium in this energy landscape the quickest.
Bechhoefer and Kumar found that beads that began at higher temperatures reached equilibrium more swiftly than those that started at lower temperatures. This is fascinating, but what does reaching equilibrium have to do with freezing?
The link between the two comes from earlier work by Zhiyue Lu from the University of North Carolina and Oren Raz from the Weizmann Institute of Science in Israel. Their paper, "Nonequilibrium thermodynamics of the Markovian Mpemba effect and its inverse," published in the Proceedings of the National Academy of Sciences (PNAS) and discussed by Quanta Magazine, suggests that hotter systems might bypass certain stages in reaching equilibrium, thus arriving at a stable state more quickly than cooler systems.
If reaching equilibrium is a crucial aspect of the freezing process of water, then the collective research of Bechhoefer, Kumar, Lu, and Raz could provide evidence supporting the Mpemba effect.
Is the Mpemba Effect a Proven Scientific Fact?
The Mpemba effect has not been universally recognized as a validated scientific phenomenon. However, extensive observations over the years, combined with recent work by Bechhoefer, Kumar, Lu, and Raz, have persuaded numerous physicists that, under certain conditions, hot water can indeed freeze faster than cold water.
Some researchers, including Harry Burridge and Paul Linden, remain doubtful. They admit that hot water may freeze faster than cold water in some situations, but even the smallest change in conditions can negate the effect. Burridge and Linden's 2016 study, "Questioning the Mpemba effect: hot water does not cool more quickly than cold," found that the occurrence of the Mpemba effect was dependent on factors like the size of the water vessel and thermometer placement. In another study, researcher James Brownridge discovered that impurities in water (as seen in Mpemba’s ice cream experiment) can alter the freezing point. While they acknowledge the possibility of hot water freezing faster in certain conditions, they argue that the effect does not consistently manifest in nature.
On the other hand, physicists such as Raúl Rica Alarcón from Spain’s University of Granada argue that recent datasets, including those by Bechhoefer and Kumar, are noteworthy. "I believe the Mpemba effect can occur under specific conditions," says Rica Alarcón, "but we are still working to determine the exact minimal conditions required for it to happen."
The Mpemba Effect in Context
Rica Alarcón observes that the Mpemba Effect is consistently linked to extreme temperature differences between the water and its environment. He further notes that equally captivating phenomena arise when these temperatures are reversed, such as placing frozen ice into a hot surrounding.
The Mpemba Effect, according to Rica Alarcón, "appears to be part of a broader set of unusual thermalization effects, occurring when a system is abruptly exposed to a thermal bath at a different temperature." It involves a phase change from hot to cold, like when you place a hot drink into the fridge or freezer. However, cold-to-hot transitions can also produce remarkable outcomes. As Rica Alarcón explains, "Fascinating phenomena occur when cold objects are rapidly heated, like when you drop an ice cube into boiling water."
Over the years, many scientists have witnessed hot water freezing at an unexpectedly fast rate. Rica Alarcón encourages us to view this phenomenon in a broader context, urging us to consider the Mpemba Effect as part of a larger scientific occurrence. He clarifies that "Thermalization processes can follow unexpected patterns because they often occur out of equilibrium," which helps explain this intriguing effect.
 We decided to give it another attempt.
Scalia Media/Shutterstock
We decided to give it another attempt.
Scalia Media/ShutterstockJust as fresh water can freeze, so too can ocean water — but at a slightly lower temperature. Due to its salt content, seawater freezes at around 28.4 degrees Fahrenheit (minus 2 degrees Celsius), whereas fresh water freezes at 32 degrees Fahrenheit (0 degrees Celsius).
