 Fire has played an undeniable role in shaping human history. It has been both a destructive force and a crucial tool in our development. It has the power to completely obliterate homes, possessions, and vast landscapes in a matter of moments. On the other hand, it has fueled our survival and progress, enabling us to cook, create, and innovate. It is a force that continuously influences our lives.
Fire has played an undeniable role in shaping human history. It has been both a destructive force and a crucial tool in our development. It has the power to completely obliterate homes, possessions, and vast landscapes in a matter of moments. On the other hand, it has fueled our survival and progress, enabling us to cook, create, and innovate. It is a force that continuously influences our lives.With the devastating capacity to burn entire forests to the ground or destroy everything in its path, fire is both a formidable weapon and a dangerous threat to humanity. Every year, it claims more lives than any other natural disaster. Yet, its destructive power is matched only by the benefits it provides. Fire's impact on human progress cannot be overstated.
At the same time, fire has proven itself invaluable. It provided early humans with light and warmth, and it enabled essential activities such as cooking, forging tools, creating pottery, and even powering machines. Fire has been both a force of destruction and one of the greatest advancements in human history, fundamentally shaping civilization. So, what is fire, precisely?
The ancient Greeks regarded fire as one of the fundamental elements of the universe, along with water, earth, and air. This classification holds up even today: like the other elements, fire can be felt, seen, and smelled. It is dynamic, portable, and powerful, capable of being transported from one place to another with ease.
Fire is fundamentally different from other elements like earth, water, and air. While these are forms of matter made up of countless atoms bonded together, fire is not considered matter. Instead, fire is a visible and tangible result of matter undergoing change – a byproduct of a chemical reaction.
Next, we will explore how this chemical reaction generates both heat and light.
What Is Fire?
 What exactly are those flickering orange flames?
Rolfo Rolf Brenner/Getty Images
What exactly are those flickering orange flames?
Rolfo Rolf Brenner/Getty ImagesFire is the result of a chemical reaction between atmospheric oxygen and a fuel source such as wood or gasoline. However, these materials won’t spontaneously combust merely by being surrounded by oxygen. To trigger the combustion, the fuel needs to be heated to its ignition temperature.
Here’s how a typical wood fire unfolds:
A heat source raises the wood to an extremely high temperature. This heat can come from various sources—such as a match, focused light, friction, lightning, or even something already on fire...
Once the wood reaches around 300°F (150°C), the heat causes some of its cellulose to break down.
The breakdown of cellulose produces volatile gases that form what we know as smoke. Smoke is composed of hydrogen, carbon, and oxygen compounds. The remaining material turns into char, almost entirely carbon, and ash, which consists of the unburned minerals in the wood like calcium and potassium. Char is the product you purchase when you buy charcoal, which is essentially wood that’s been heated to drive out the volatile gases, leaving behind carbon. This is why charcoal burns with no smoke.
The actual combustion of wood occurs in two distinct reactions:

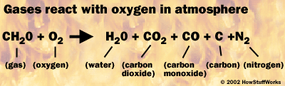
- Once the volatile gases reach a high enough temperature (around 500°F or 260°C for wood), the molecules break apart and their atoms react with oxygen, creating water, carbon dioxide, and other byproducts. In other words, the gases burn.
- The carbon in the char also reacts with oxygen, but at a much slower rate. This slower process explains why charcoal in a BBQ stays hot for a long time.
One consequence of these chemical reactions is the generation of a significant amount of heat. This heat sustains the fire. Some fuels, like gasoline, burn in one step. The heat causes gasoline to vaporize, and the gas burns completely. No char remains. Humans have also figured out how to control the fuel and manage a fire. A candle is an example of a tool that gradually vaporizes and burns wax.
As the materials heat up, the rising carbon atoms (along with atoms from other substances) release light. This phenomenon, where heat generates light, is known as incandescence, and it's the same principle behind the light in a light bulb. It’s also what creates the visible flame. The color of the flame depends on the material being burned and its temperature. The hottest part of the flame, typically at the base, appears blue, while the cooler parts at the top glow orange or yellow due to uneven temperature distribution.
In addition to emitting light, the rising carbon particles can settle on nearby surfaces, forming soot.
 In microgravity, fire takes on a spherical shape.
Photo courtesy of NASA
In microgravity, fire takes on a spherical shape.
Photo courtesy of NASAWhat makes fire dangerous is the fact that the chemical reactions involved are self-sustaining. The heat generated by the flame maintains the fuel at its ignition temperature, allowing it to continue burning as long as there is fuel and oxygen. The flame also heats nearby fuel, causing it to release gases. When these gases are ignited, the fire spreads.
On Earth, gravity influences how flames burn. The hot gases in the flame are much hotter (and less dense) than the surrounding air, causing them to rise towards areas of lower pressure. This explains why fire typically moves upward and why flames always seem to point upward. However, in a microgravity environment, such as aboard the space shuttle, the fire would form a sphere instead!
Fire Variables
In the previous section, we learned that fire is the result of a chemical reaction between two gases—usually oxygen and a fuel gas. The heat creates the fuel gas. Essentially, with the energy provided by heat, atoms in one gaseous compound break their bonds and combine with oxygen atoms in the air, forming new compounds and releasing more heat.
Not all compounds will easily break apart and recombine in this way – the atoms must be attracted to each other in just the right way. For instance, when water is boiled, it turns into steam, but this gas doesn’t react with the oxygen in the air. The attraction between the hydrogen and oxygen atoms in a water molecule isn’t strong enough to cause a reaction with the oxygen molecules in the air, preventing the water from breaking apart and recombining.
The most combustible compounds are those that contain carbon and hydrogen, as these elements can easily recombine with oxygen to form carbon dioxide, water, and other gases.
Different fuels ignite at different temperatures. To turn any material into a gas, a specific amount of heat energy is required, and even more heat is needed to trigger the reaction with oxygen. The necessary heat varies depending on the molecular structure of the fuel. A fuel’s piloted ignition temperature is the heat level required to create a gas that will ignite when exposed to a spark. In contrast, the unpiloted ignition temperature is much higher, and the fuel will ignite even without a spark.
The size of the fuel also influences how easily it catches fire. A larger fuel, like a thick tree, can absorb a great deal of heat, so it requires much more energy to bring any part of it up to its ignition temperature. On the other hand, a toothpick ignites more easily because it heats up very quickly.
A fuel’s heat production depends on how much energy the gases release during combustion and how quickly the fuel burns. Both of these factors are primarily determined by the fuel’s composition. Some compounds react with oxygen to produce a significant amount of "extra heat energy," while others release less. Similarly, the fuel’s reaction with oxygen can occur rapidly or more gradually.
The shape of the fuel also plays a role in how quickly it burns. Thin pieces of fuel combust faster than larger chunks because a larger portion of their mass is exposed to oxygen at any given time. For example, you could burn a pile of wood splinters or paper far more quickly than a solid block of wood of the same weight, since splinters and paper have a much higher surface area.
In this way, fires from different types of fuel are like distinct animal species -- each behaves in its own unique way. Experts can often determine the origin of a fire by examining the damage it caused to the surroundings. A fire fueled by fast-burning materials that produce intense heat will cause a different kind of damage compared to one fueled by slow-burning materials that generate less heat.
For a deeper understanding of fire science, explore the links provided below.
