 Desalination facilities, such as the largest one in Europe located in El Prat del Llobregat near Barcelona, employ reverse osmosis to transform salty seawater into potable water. This image from Nov. 28, 2022, captures the inner workings of the plant. JOSEP LAGO/AFP/Getty Images
Desalination facilities, such as the largest one in Europe located in El Prat del Llobregat near Barcelona, employ reverse osmosis to transform salty seawater into potable water. This image from Nov. 28, 2022, captures the inner workings of the plant. JOSEP LAGO/AFP/Getty ImagesIf you've ever seen a news report on a severe drought and then looked at your beach-themed screensaver, you may have wondered, 'Why not just use the ocean water?'
Naturally, you might have considered a few things. First, the ocean is salty. Second, salty water isn't ideal for drinking or agriculture. Third, removing salt from water may seem impossible—just like you can't simply remove sugar from tea. Or can you?
Reverse osmosis is a key process in desalination, the technique used to remove salt from seawater. Beyond desalination, reverse osmosis is also applied in recycling, wastewater treatment, and even energy production.
Water scarcity is rapidly becoming a critical global issue. With climate change, we face extreme weather patterns, including severe flooding in some areas, droughts in others, and fluctuating sea levels. On top of this, overpopulation and the environmental strain it brings make water a key environmental challenge for the coming generations.
Reverse osmosis is being adapted by water treatment plants to address some of these pressing issues. In Perth, Australia—dry and arid, yet surrounded by ocean—almost 17% of the city's drinking water comes from desalinated seawater processed by a reverse osmosis plant [source: ExcelCalcs]. Worldwide, over 22,757 desalination plants now operate, according to the International Desalination Association.
While understanding how reverse osmosis works to convert seawater into drinkable water is valuable, what we really need to grasp is the process itself. If you're already familiar with the concept of 'reverse,' let's first examine how osmosis works before we combine the two concepts.
What is Osmosis?
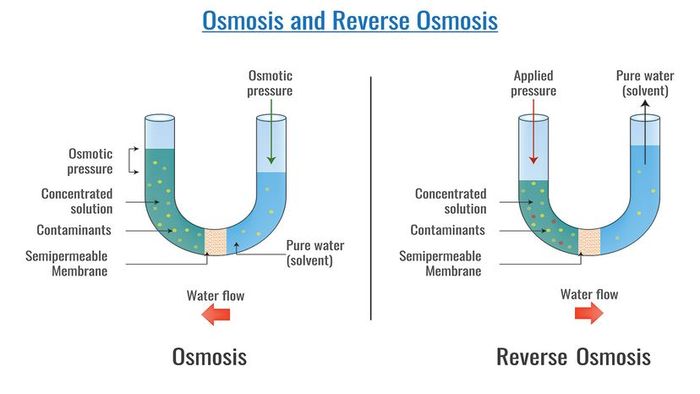 Osmosis is the movement of fluid through a semipermeable membrane. Reverse osmosis works as a water purification method by moving water from a high-concentration solution to a lower-concentration one. Nandalal Sarkar/Shutterstock
Osmosis is the movement of fluid through a semipermeable membrane. Reverse osmosis works as a water purification method by moving water from a high-concentration solution to a lower-concentration one. Nandalal Sarkar/ShutterstockOsmosis refers to the movement or diffusion of water and other solvents through a semipermeable membrane that prevents the passage of dissolved solutes [source: Encyclopedia Britannica].
Not sure you get it? Don't worry, most people don't, which is why there are so many explanations and analogies out there to help clarify osmosis. We'll explore a few, but first, let's break it down into its fundamental components so we can get a better understanding.
First, let's prepare our solution. Start with a simple cup of water. To make things more interesting, we'll call the water the 'solvent'—since that's exactly what it is. Now, to make it more flavorful, we'll add some sugar. The sugar becomes the 'solute.' So, we now have water (the solvent) mixed with sugar (the solute), which creates a sugary solution.
Now that we have our sugary water solution, let's take a U-tube. This isn't an internet video of kittens and monkeys hugging, but rather a U-shaped beaker. In the middle of the tube, imagine a piece of Gore-tex dividing it in half. Gore-tex serves as our 'semipermeable membrane.' It's a thin plastic with millions of tiny pores that let water vapor pass but block liquid. (Unlike Saran wrap, which would prevent anything from passing, or cotton fabric, which would let almost everything through.)
In one side of the U-tube, we pour our sugar water solution, and in the other, we pour plain water. This is when the magic of osmosis starts—if you find water moving around magical. The level of liquid on the sugar water side will gradually rise as the solvent (water) moves through the Gore-tex, aiming to balance the sugar-to-water ratio on both sides.
So why does this happen? Simply put, water seeks balance. Since the sugar side is more concentrated, pure water from the other side moves over to equalize the concentrations, or until osmotic pressure (the force created by the molecules moving) is reached [source: Encyclopedia Britannica].
And there you have it: osmosis is when a solvent from a lower concentration solution moves through a membrane to a higher concentration solution, effectively diluting it. You got it!
Now that we've shown how osmosis naturally flows in one direction, let's turn everything on its head and reverse the process.
Reverse Osmosis: Flip It
Freddie Mercury and David Bowie both knew that pressure could burn down buildings, tear families apart, put people on the streets—and also create an incredibly catchy tune. What they missed? That pressure is also what drives reverse osmosis.
We’ve learned that in osmosis, a lower-concentrated solution pushes its solvent towards the higher-concentrated solution. In reverse osmosis, we flip the process: the solvent moves from a high-concentration solution to a low-concentration one. Instead of equalizing the solute-to-solvent balance, it separates the solute from the solvent.
But as we’ve seen, solutions don’t naturally want to do this. So, how do we make reverse osmosis happen? Like Freddie and Bowie, we apply pressure. Let’s take saltwater as an example:
In reverse osmosis, we place saltwater on one side of a tank and pure water on the other, separated by a semipermeable membrane. By applying enough pressure on the saltwater side to counteract the osmotic pressure from the pure water side, we push the saltwater through the membrane. This requires about 50-60 bars of pressure [source: Lenntech]. Due to the size of the salt molecules, only the smaller water molecules can pass through, leaving fresh water on the pure water side and the salt behind.
Reverse osmosis occurs when pressure is applied to a concentrated solution, forcing the solvent to pass through a membrane to the less concentrated side, leaving behind a higher concentration of solute on one side and pure solvent on the other.
While it's impressive to explain reverse osmosis at a dinner gathering, its real-world applications are far more intriguing and can spark even more interesting discussions.
Applications of Reverse Osmosis
 In 2021, E-cells were utilized in an industrial reverse osmosis water treatment system in Penang, Malaysia, as documented by Mohd Rodi/Shutterstock.
In 2021, E-cells were utilized in an industrial reverse osmosis water treatment system in Penang, Malaysia, as documented by Mohd Rodi/Shutterstock.Unlike regular osmosis, reverse osmosis isn’t something we can often witness in our daily lives. It wasn’t until the 1950s that researchers began investigating reverse osmosis as a method for desalinating ocean water. They found that by applying pressure to saltwater, they could create fresh water, but the amount was so small it wasn’t practical. What changed?
A breakthrough came with a more sophisticated filter developed by two UCLA scientists. They created hand-cast membranes made from cellular acetate (the same material used in photographic film). This innovation allowed significantly larger quantities of water to pass through much more quickly, and in 1965, the first reverse osmosis desalination plant started a small-scale operation in Coalinga, California [source: The Economist].
One of the most common applications of reverse osmosis, which we’ve already touched on, is water desalination. This includes large-scale facilities (over 177 countries use desalination), as well as smaller operations, such as portable filters you might use when camping to ensure access to clean drinking water [source: FDU].
Reverse osmosis is also one of the few effective methods for removing certain minerals or chemicals from water supplies. Some sources have dangerously high levels of natural fluoride, which can cause enamel fluorosis (mottled teeth) or even skeletal fluorosis (the bending of bones). Reverse osmosis can filter out fluoride and other contaminants on a large scale, a task beyond the capabilities of standard charcoal filters commonly used in homes.
It’s also a key technology in recycling efforts. For example, the chemicals used in metal treatment create toxic wastewater, but reverse osmosis can extract clean water for safer chemical disposal. Even more fascinating is its use in wastewater treatment, where it purifies wastewater to make it drinkable. This process, dubbed "toilet to tap," may sound unsettling, but it offers a promising solution for developing nations in need of potable water.
Reverse osmosis isn't just for water purification—it's used across various industries. For example, maple syrup production uses osmosis to separate the sugary concentrate from the water in sap. The dairy industry relies on reverse osmosis filtration to concentrate whey and milk, while the wine industry uses it to remove unwanted elements like certain acids, smoke, or to regulate alcohol content. It’s also key in producing pure, contaminant-free ethanol.
One intriguing feature of reverse osmosis is that the high pressure involved can actually be recycled. Water is pushed through by high-pressure pumps, and the leftover salty water is expelled at a very high rate. By channeling this discharge through a turbine or motor, the pressure can be harnessed and used to power the original pumps, essentially harvesting energy in a closed loop.
While the industrial applications are impressive, you might be wondering: how does reverse osmosis affect you, the consumer, on a smaller scale?
Smaller-scale Applications of Reverse Osmosis
 On July 13, 2020, an employee of Berliner Wasserbetriebe in Berlin pours drinking water from a jug. The water comes from a reverse osmosis plant that provides clean drinking water for over a million Berlin residents. Jens Kalaene/picture alliance/Getty Images
On July 13, 2020, an employee of Berliner Wasserbetriebe in Berlin pours drinking water from a jug. The water comes from a reverse osmosis plant that provides clean drinking water for over a million Berlin residents. Jens Kalaene/picture alliance/Getty ImagesIf you’re craving some fresh reverse osmosis water, why not simply fill up a pitcher with it and enjoy a refreshing drink? But keep in mind, it's not as straightforward as it seems.
It’s not that simple, though. Reverse osmosis requires significant pressure, so you won’t find a pitcher designed for it. If you want reverse osmosis water flowing throughout your entire home, it means investing in a whole new water system. However, if you just need it for drinking or cooking, you won’t need to turn your basement into a mini reverse osmosis plant.
Your first option for a smaller-scale system is an 'under the counter' setup. A reverse osmosis system connects to your water supply under the sink, where water is filtered through multiple stages to ensure purity. The purified water is then stored in a tank beneath the sink, with a dedicated faucet installed on the countertop. Such systems typically cost between $200 and $500, and installation is often a DIY job, so it’s important to feel confident in your repair skills. [source: Fitzgerald]
For those who are not comfortable installing a faucet or water system (or perhaps worried that your landlord might not approve of your DIY efforts), there are reverse osmosis countertop filters. These compact systems hook directly to your faucet, allowing you to filter the water through a small device that fits beside your microwave. The purified water can then be easily poured into a pitcher for convenient access.
However, countertop systems might not be the best option for everyone. They tend to be slower due to the low-flow faucets, and they typically cost around $150 or more. Additionally, the filters need to be replaced every few months, which adds another cost of about $30 each time. [source: Fitzgerald]
Disadvantages of Reverse Osmosis
Now that we've explored some of the ways reverse osmosis can be beneficial, it’s worth asking: Is reversing nature really a good idea? There are some challenges that arise from reverse osmosis, and we'll begin by looking into what happens during desalination using reverse osmosis.
After filtering the water, you're left with clean drinking water, but there's also the issue of dealing with the leftover brine, which is typically twice as salty as seawater. [source: The Economist] What should be done with this brine? Is it problematic to release it back into the ocean? According to the Australian Centre for Water research, salinity levels seem to normalize at around 1,600 feet (500 meters) from the discharge point. [source: The Economist] However, the impact of metals and chemicals trapped in the brine remains unclear and needs further investigation.
A significant issue with reverse osmosis systems is the amount of water wasted during the process. Studies indicate that some reverse osmosis systems can waste between 3 and 20 times the amount of water they actually produce.
Reverse osmosis systems, in general, are also not entirely self-sustaining. Water must be pretreated with chemicals, for instance, so nothing will clog the fine membrane. And the membrane itself is not entirely easy to deal with; it must be cleaned often, and can trap bacteria. A concern unique to the desalination plants is that small fish or marine life can be sucked into the system; adjusting intake pressures and velocities can usually prevent harm.
The biggest impediment of reverse osmosis filtration systems is the cost.
Depending on its design, a typical ocean water desalination plant that processes 20 million gallons (76 liters) of seawater daily could cost as much as $514 to $740 million to build and require nearly $20 million per year to operate.
For a developing nation, installing reverse osmosis systems is a fairly impractical possibility. Organizations like the WHO and UNICEF consider building reverse osmosis water treatment plants — to remove toxins or provide a clean water supply — part of their mission.
Thinking you need some reverse osmosis water for your beloved guppies? You might want to think again. While reverse osmosis units can certainly filter out a lot of harmful impurities, you'll also need to add some essential minerals back in that get taken out in the process. Make sure to research what is taken out of water in reverse osmosis, and what minerals your fish need to thrive [source: Foster and Smith Aquatics].
