
Essential Insights
- Artificial sweeteners provide a calorie-conscious alternative to sugar, though some still contain calories.
- While regulatory bodies like the FDA and manufacturers claim these sweeteners are safe, certain health professionals and advocacy groups raise concerns about potential links to cancer and neurological disorders.
- Research indicates that artificial sweeteners might interfere with the body's ability to gauge fullness, possibly resulting in higher calorie intake.
Cutting out sugar is a highly effective strategy for reducing calorie consumption when aiming to lose weight. Many people turn to artificial sweeteners and sugar-free products to satisfy their sweet tooth without the extra calories. However, not all artificial sweeteners are devoid of calories—some contain nearly as many as regular sugar. Additionally, their safety remains a topic of debate. While the Food and Drug Administration (FDA) and manufacturers affirm their safety, some consumer organizations and medical experts remain skeptical.
Even if you consciously avoid artificial sweeteners, they might still be present in unexpected everyday items. For instance, did you realize that toothpaste, mouthwash, chewable vitamins, and cough drops often contain these sweeteners?
This article explores the origins of artificial sweeteners, their applications, and the regulatory approval process. Additionally, it delves into the specifics of various sweeteners and the debates surrounding their use.
Sweet Thing
 ®
®Sweetness isn't exclusive to sugar; numerous organic, synthetic, and inorganic compounds can impart a sweet taste. Many plants contain sugar-based compounds called glycosides. For instance, stevia, a plant rich in glycosides, has been utilized for hundreds of years to sweeten foods and beverages.
The sweetness we experience is determined by the interaction between molecules and the receptors on our tongue. A stronger interaction results in a sweeter taste. (Explore this animation to understand how taste buds function.)
Researchers at Senomyx have pinpointed the specific taste bud receptor responsible for detecting sweetness. Both sugar and artificial sweeteners bind to this receptor, triggering the sweet sensation we associate with them. These receptors are located on the surfaces of cells throughout the tongue and mouth, sending signals to the brain to indicate sweetness.
Artificial sweeteners are substances designed to mimic the sweet taste of sugar. Some are low-calorie because they are significantly sweeter than sugar, requiring only small amounts. Others are low or no-calorie because the body cannot process them, allowing them to pass through the digestive system without being absorbed.
Next, we’ll explore the history and applications of artificial sweeteners.
is a natural sweetener derived from a plant native to Brazil and Paraguay, used for centuries in various countries. It is approximately 300 times sweeter than sugar and contains no calories since the body does not metabolize it.
While supporters cite studies showing no negative effects, other research suggests it may reduce sperm production and result in fewer and smaller offspring. The FDA has not approved it for food use, but it is available as a dietary supplement.
Artificial Sweetener Basics

Saccharin, the first artificial sweetener, was accidentally discovered in 1879 by a scientist who neglected to wash his hands before eating and noticed a sweet taste on his fingers. Similarly, other artificial sweeteners were found when researchers licked their fingers during experiments or smoked cigarettes near sweet compounds. Ironically, lapses in personal hygiene have greatly benefited the diet industry.
These accidental discoveries highlight the artificial nature of these sweeteners, despite marketing claims. Splenda, for instance, faced legal action from the sugar industry for misleading consumers into believing it was a natural product. A study by the Center for Science in the Public Interest (CSPI) revealed that 57% of people mistakenly believed Splenda was natural [ref].
The variety of artificial sweeteners exists because no single sweetener suits every application. Sucralose (Splenda) is ideal for baking due to its heat resistance, while aspartame is commonly used in "light" dairy products like yogurt. Sugar alcohols, such as xylitol and sorbitol, are popular in sugar-free ice cream. However, while they don’t spike blood sugar or cause tooth decay, they contain nearly as many calories as sugar.
Beyond "light" and "sugar-free" foods, artificial sweeteners are found in liquid and chewable medications, throat lozenges, cough drops, vitamins, toothpaste, and mouthwash—products that benefit from sweetness without sugar. Some manufacturers use artificial sweeteners simply because they are cheaper. A UK Food Commission report revealed that certain non-diet orange sodas used artificial sweetener blends, as aspartame costs just two cents per liter compared to six cents for sugar.
To avoid consuming artificial sweeteners, carefully inspect ingredient labels and familiarize yourself with their scientific names rather than just their brand names.
Next, we’ll explore the debates and controversies surrounding artificial sweeteners.
Researchers at Senomyx are working to identify chemicals that amplify the effectiveness of sweetness receptors. Their lab contains artificial taste buds that emit a green glow upon contact with sugars. By testing various chemicals using these "taste buds," they aim to discover a compound that mimics sugar’s taste without the aftertaste—and ideally, without adverse effects.
Artificial Sweetener Controversy

Concerns about artificial sweeteners emerged soon after they entered the food market. When public health issues, such as a rise in specific types of cancer, arise, scientists investigate newly introduced substances. Saccharin and cyclamate, the oldest artificial sweeteners, were both linked to cancer in lab mice and rats. Studies connecting saccharin to bladder cancer may have fueled the widespread belief that all artificial sweeteners are carcinogenic. The media is often criticized for reporting potential cancer risks without sufficient scientific evidence.
Beyond cancer, numerous websites and forums discuss various risks and side effects linked to aspartame, sucralose, and other artificial sweeteners. Conditions like multiple sclerosis, Alzheimer's disease, brain tumors, and nervous disorders have been attributed to their use. While aspartame faces the most criticism, sucralose is also being closely examined.
The FDA continues to support the safety of the products it has approved. A Q&A section on the FDA’s website states:
Health organizations like the American Heart Association and the American Diabetes Association endorse the use of artificial sweeteners for diabetics and weight management.
Despite the widespread availability of diet sodas and sugar-free foods, obesity remains a significant issue. A Purdue University study suggests that diet soft drinks might contribute to the problem. Researchers in the Department of Psychological Sciences found that artificial sweeteners could disrupt the body’s natural calorie-counting mechanism. Our bodies gauge calorie intake partly based on sweetness—the sweeter and denser a food, the higher its calorie content. This mechanism helps signal when to stop eating, but artificial sweeteners may interfere with this process.
Artificial sweeteners disrupt the body’s natural calorie estimation process. Consuming foods and drinks with artificial sweeteners, which are lower in calories, may train the body to no longer link sweetness with high-calorie content. As a result, when consuming sugar-sweetened items, the body misjudges the calorie count, leading to overconsumption.
In the Purdue study, rats exposed to both artificially-sweetened and sugar-sweetened liquids for 10 days consumed more of a high-calorie chocolate snack compared to rats given only sugar-sweetened liquids. The inconsistent association between sweetness and calories impaired their ability to regulate calorie intake [ref].
The National Soft Drink Association countered that the study was flawed, citing numerous other studies demonstrating that low-calorie sweeteners aid in weight loss.
In the next section, we’ll explore how artificial sweeteners enter the food supply chain.
From Lab to Kitchen
 Photo courtesy Office of NIH History
Photo courtesy Office of NIH HistoryThe Delaney Clause, part of the Food Additives Amendment of 1958, mandates that no food additive can be deemed safe if it causes cancer in humans or animals. This is tested by administering high doses of the additive, such as artificial sweeteners, to a limited number of rats and mice.
High doses are used to account for the small sample size of animals, typically a few hundred, and the possibility that rodents may be less sensitive to the chemical than humans. While it might seem that only large doses cause cancer, smaller doses can also be carcinogenic, albeit less frequently.
Companies seeking to market a new sweetener or food additive must petition the FDA for approval, unless the additive is composed of ingredients generally recognized as safe (GRAS). The FDA typically requires robust evidence, including animal studies, to ensure the additive poses no harm even if consumed in unexpectedly large amounts. Human studies may also be submitted.
The FDA evaluates an additive’s composition and properties during the approval process. Key considerations include whether the sweetener breaks down into harmful by-products, the expected consumption levels, and potential long-term effects. Since absolute safety cannot be guaranteed, the FDA bases its decision on the best available scientific evidence.
Once a sweetener is approved, the FDA specifies the food categories it can be used in, the maximum allowable amounts, and its labeling requirements. It is then classified as GRAS. The FDA continuously monitors consumption and new safety research to ensure the sweetener remains within safe usage limits.
The Adverse Reaction Monitoring System (ARMS) acts as a continuous safety mechanism for all food additives. It tracks and investigates complaints linked to specific additives. If adverse reactions pose a public health risk, the FDA will intervene.
Next, we’ll explore some artificial sweeteners in detail, starting with saccharin.
The 1958 Food Additives Amendment to the Federal Food, Drug, and Cosmetic Act (FD&C Act) establishes four legal categories for substances added to foods:
- Food additives: Substances without a proven safety record must undergo FDA review and approval before use in food products.
- Generally recognized as safe (GRAS): Substances with a proven safety record, either through pre-1958 use or scientific evidence, do not require FDA approval.
- Prior-sanctioned: Substances deemed safe by the FDA or USDA before 1958 for specific foods.
- Color additives: Dyes used in foods, drugs, cosmetics, and medical devices must be FDA-approved.
Saccharin
 Tab is among the limited number of products available today that include saccharin.
Tab is among the limited number of products available today that include saccharin.Basics
Saccharin, derived from the Latin word for sugar, is a synthetic compound discovered in 1879, marking it as the first artificial sweetener. It was accidentally found by two chemists at Johns Hopkins University while working on coal tar derivatives for chemical dyes. One chemist, neglecting to wash his hands, noticed a sweet taste on his fingers. Saccharin is 300 times sweeter than sugar and is not metabolized, making it calorie-free.
Use
Today, saccharin is found in very few products. Fountain Diet Coke and Pepsi use a combination of saccharin and aspartame, as does Tab. It is also sold as Sweet 'N Low, a tabletop sweetener in the iconic pink packets.
Controversy
Saccharin has a contentious history. In 1977, the FDA attempted to ban it due to animal studies linking it to cancer, particularly bladder, uterine, ovarian, and skin cancers. However, pressure from the diet food industry and consumers kept it on the market, albeit with a warning label indicating it caused cancer in lab animals. This label remained until the late 1990s. The Calorie Control Council argued that humans do not develop bladder cancer in the same way as rats, advocating for the removal of the warning.
In 2000, the National Cancer Institute (NCI) found no increased bladder cancer risk among saccharin users compared to the general population. However, heavy users (six or more servings of saccharin or two or more 8-ounce diet drinks daily) showed some evidence of elevated risk, especially from tabletop sweeteners or diet sodas. Based on this and other animal studies, saccharin was deemed not a significant risk factor for human bladder cancer. It was removed from the NIH’s carcinogen list, and Congress eliminated the warning label requirement.
Next, we’ll explore aspartame, another artificial sweetener surrounded by controversy.
was discovered accidentally in 1937 by a University of Illinois graduate student. It is 30 to 50 times sweeter than sugar. The FDA banned cyclamate in 1970 after animal studies linked it to cancer, but a petition for its re-approval is currently under review.
is still used in over 50 countries, including Canada, where it is the sweetener in Sweet n' Low. Ironically, saccharin is banned in Canada except for diabetic use.
Aspartame

Basics
Aspartame, marketed under brands like Equal, NutraSweet, and NatraTaste, is made from two amino acids: aspartic acid and phenylalanine. It was discovered in 1965 by chemist Jim Schlatter at G. D. Searle & Co. (now part of Pfizer) while he was testing an anti-ulcer drug. Schlatter accidentally tasted it when he licked his finger to pick up a piece of paper.
Aspartame is 180 to 200 times sweeter than sugar, requiring only minimal amounts to sweeten food or drinks. Upon digestion, it breaks down into aspartic acid, phenylalanine, and methanol (wood alcohol).
Individuals with the rare genetic condition phenylketonuria (PKU) must avoid aspartame due to their inability to metabolize phenylalanine. Consuming phenylalanine-rich foods or drinks can lead to its accumulation, potentially causing mental impairment or brain damage. Newborns are routinely screened for PKU.
Use
Aspartame is not typically used in baked or heated foods because it breaks down under heat. According to the official aspartame Web site, it is found in over 6,000 products, including carbonated and powdered soft drinks, chewing gum, candies, gelatins, dessert mixes, puddings, frozen desserts, yogurt, tabletop sweeteners, and some pharmaceuticals like vitamins and sugar-free cough drops. The FDA approved aspartame as a food additive in 1981.
Controversy
Aspartame has sparked more controversy than any other artificial sweetener. Since its approval, 75% of all complaints reported to ARMS have been related to aspartame. However, only about 1% of people who experience issues actually report them [ref].
Although no officially recognized studies have confirmed issues with aspartame, many consumer groups and individuals claim it causes health problems such as headaches, seizures, chronic fatigue syndrome, memory loss, and dizziness. It has also been linked to an increased risk of multiple sclerosis, Alzheimer's disease, and cancer.
Critics argue that while the amino acids in aspartame are naturally present in our diets, they are typically consumed alongside other amino acids that neutralize potential negative effects. When isolated, as in aspartame, there is concern that these amino acids may enter the central nervous system in unusually high concentrations.
A similar issue is alleged with methanol, a byproduct of aspartame digestion. Naturally occurring methanol in fruit juices and alcoholic beverages is accompanied by ethanol, which counteracts methanol toxicity. However, 10% of aspartame is absorbed as methanol. The EPA recommends a daily limit of 7.8 milligrams of methanol, yet a one-liter aspartame-sweetened drink contains about 56 milligrams—eight times the recommended amount.
In 2005, the European Journal of Oncology published a study linking aspartame to lymphomas and leukemia in female rats. The lowest dose associated with increased risks was equivalent to eight cans of aspartame-sweetened soda daily for adults (two for children). Brain tumors were also found in 12 of 1500 aspartame-treated animals, while none were found in the control group.
Conversely, numerous studies conducted by various groups have concluded that aspartame is safe and poses no health risks.
Next, we’ll examine acesulfame, a less commonly known sweetener.
is structurally similar to aspartame, as it is derived from aspartic acid and phenylalanine. However, it is more stable due to a stronger bond between the amino acids. Developed over two decades, it is 8,000 times sweeter than sugar. According to the neotame Web site, just six milligrams can sweeten a 12-ounce drink.
Unlike aspartame, neotame does not break down into phenylalanine, making it safe for individuals with PKU. It is also suitable for cooking and baking.
was approved by the FDA in 2002 for use in beverages, frozen desserts, yogurt, ice cream, chewing gum, candy, baked goods, toppings, fruit spreads, breakfast cereals, and more. However, due to its recent introduction, few products currently use neotame.
Acesulfame
 This meal replacement powder incorporates acesulfame as its sweetening agent.
This meal replacement powder incorporates acesulfame as its sweetening agent.Basics
Acesulfame, also known as acesulfame potassium or acesulfame K, is a synthetic compound approximately 200 times sweeter than sugar. Discovered in 1967 by Hoechst AG, a German life-sciences company now part of Aventis, it is not metabolized by the body, making it low in calories. It is produced through a process involving acetoacetic acid and potassium.
Use
Acesulfame received FDA approval in 1988 and is now used in over 4,000 products globally. In the U.S., it is approved for candies, tabletop sweeteners, chewing gums, beverages, dessert and dairy mixes, baked goods, alcoholic drinks, syrups, frozen desserts, and sweet toppings. Marketed under brands like Sweet One and Sunett, it is often blended with other sweeteners to mimic the taste of sugar.
Controversy
In August 1988, the Center for Science in the Public Interest (CSPI) requested a halt to acesulfame's FDA approval, citing "significant doubt" about its safety. CSPI argued that the studies were flawed and failed to adequately prove acesulfame's non-carcinogenic properties. According to CSPI, "...acetoacetamide, a breakdown product of acesulfame, has been shown to affect the thyroid in rats, rabbits, and dogs. Feeding rats 1% and 5% acetoacetamide for three months caused benign thyroid tumors. The rapid tumor development raises serious concerns about its carcinogenic potential" [ref].
Next, we’ll explore one of the newest artificial sweeteners: sucralose.
Sucralose
Basics
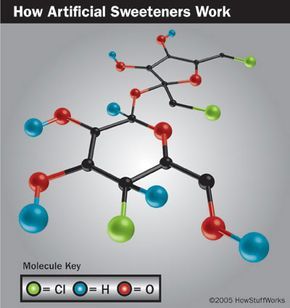
Sucralose is made by replacing three hydrogen (H) and oxygen (O) groups in a sugar molecule with three chlorine (Cl) atoms. This transformation turns it into an artificial sweetener that is 600 times sweeter than sugar.
Since sucralose is not metabolized by the body, it contains almost no calories. The chlorine atoms, which prevent absorption, also make it heat-stable, allowing it to be used in baking—unlike other sweeteners that lose their sweetness when heated. The FDA approved sucralose for use in foods and beverages in 1998.
Use
Sucralose, sold under the brand name Splenda, is the fastest-growing artificial sweetener in the market. It is used in a wide range of products, including frozen desserts, sodas, cookies, gum, and candies. It is also available in bulk for baking and in small yellow packets for sweetening coffee or tea.
Controversy
Sucralose is less controversial than aspartame, but the FDA’s 1998 approval report noted that it is "weakly mutagenic in a mouse lymphoma mutation assay," indicating minor genetic damage in mouse cells. Additionally, one of its breakdown products was found to be "weakly mutagenic in the Ames test," a standard method for detecting potential carcinogens [ref].

Another controversy surrounds Splenda’s marketing. Its tagline, "Splenda is made from sugar, so it tastes like sugar," has led many to believe it is a natural and healthier sweetener, which is not accurate. Without long-term studies, it remains unclear whether sucralose is healthier than other artificial sweeteners.
Next, we’ll examine tagatose, another newer artificial sweetener.
Tagatose
Basics
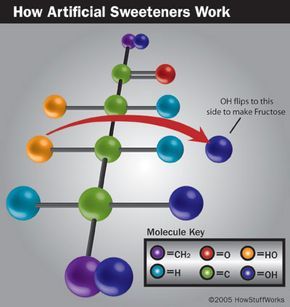
Tagatose, also known as Natrulose, is a novel artificial sweetener derived from lactulose, or milk sugar. The process involves two steps: first, lactose is hydrolyzed (broken down using water) into glucose and galactose. Second, galactose is isomerized (rearranged into a molecule with the same formula but a different structure) to form D-tagatose by adding calcium hydroxide.
Tagatose was discovered when Gilbert Levin, founder of Spherix, sought a new sweetener and theorized that a "left-handed" sugar might work. In chemistry, molecules can be chiral (either "left-handed" or "right-handed") if they are non-superimposable mirror images, like human hands. Levin was mistakenly sent D-tagatose, a "right-handed" molecule similar to L-fructose, which turned out to be exactly what he needed.
Tagatose contains about 1.5 calories per gram and has minimal effects on blood glucose or insulin levels. Since it is not digested, it passes through the body without being absorbed. It matches sugar in bulk and sweetness and can be blended with other artificial sweeteners to enhance flavor and texture. The FDA granted it Generally Recognized As Safe (GRAS) status in 2001 based on the manufacturer’s submission.
Use
Due to its recent introduction, tagatose is not yet widely used. It is currently found in Diet Pepsi Slurpees at 7-Eleven and Florida Gold’s Light and Tangy frozen juice concentrate. It is suitable for breakfast cereals, diet soft drinks, health bars, frozen yogurt, nonfat ice cream, soft and hard candies, frosting, and chewing gum.
Controversy
As tagatose is relatively new, there is little controversy surrounding it. However, it is poorly absorbed and may cause gastrointestinal discomfort if consumed in large amounts.
Next, we’ll explore sugar alcohols.
Dihydrochalcones (DHCs) are calorie-free sweeteners derived from citrus fruit bioflavonoids, while glycyrrhizin is a noncaloric extract from licorice root, 50 to 100 times sweeter than sugar. Both are already approved as flavors and flavor enhancers.
Alitame, marketed as Aclame, is reportedly 2,000 times sweeter than sugar. A petition for its approval has been submitted to the FDA.
Sugar Alcohols
 This sugar-free gum uses sorbitol, a sugar alcohol, as its sweetening agent.
This sugar-free gum uses sorbitol, a sugar alcohol, as its sweetening agent.Basics
Sugar alcohols are created by adding hydrogen atoms to sugars. They do not contain ethanol and are unrelated to alcoholic beverages. Naturally occurring in foods like fruits and berries, they have about half to one-third fewer calories than sugar because they convert to glucose more slowly. They typically do not cause blood sugar spikes, making them suitable for diabetics in moderation. However, some individuals with Type I diabetes may experience blood sugar increases if they consume large amounts.
The calorie content of sugar alcohols varies. For instance, erythritol is poorly absorbed, making it virtually calorie-free. Others, however, can have nearly as many calories as sugar, so they are not always used in "diet" foods but are common in sugar-free gum.
Use
Sugar alcohols are commonly found in sugar-free processed foods like hard candies, cookies, chewing gums, soft drinks, throat lozenges, toothpaste, and mouthwash. Check product labels for ingredients such as mannitol, sorbitol, xylitol, lactitol, isomalt, erythritol, maltitol, and hydrogenated starch hydrolysates (HSH).
Controversy
The FDA mandates that products containing a daily dose equivalent to 50 grams of sorbitol or 20 grams of mannitol include a "laxative effect" warning. This is because high levels of unabsorbed sugar alcohols in the intestines can lead to bloating, gas, and diarrhea. According to the Center for Science in the Public Interest, as little as 10 grams of sorbitol can cause gastrointestinal discomfort.
