 Lasers are not limited to games like laser tag; they are integral to many common devices, including CD players and dental tools. RichLegg/Getty Images
Lasers are not limited to games like laser tag; they are integral to many common devices, including CD players and dental tools. RichLegg/Getty ImagesFrom "Star Wars" to "Star Trek" and "Battlestar Galactica," laser technology is a cornerstone of science fiction narratives, often symbolizing advanced weaponry and futuristic spacecraft.
Lasers are also deeply embedded in our daily routines, appearing in a vast array of applications. They are essential in devices ranging from CD players to precision dental instruments and industrial cutting machines. Additionally, they are used in medical procedures such as tattoo removal, hair restoration, and eye surgeries.
What exactly defines a laser? How does a laser beam differ from the light emitted by a flashlight? More importantly, what sets laser light apart from other forms of light, and how are lasers categorized?
This article delves into the various types of lasers, their unique wavelengths, and their practical applications, such as in laser pointers. To begin, we’ll explore the foundational principles of laser technology by breaking down the structure of an atom.
The Basics of an Atom
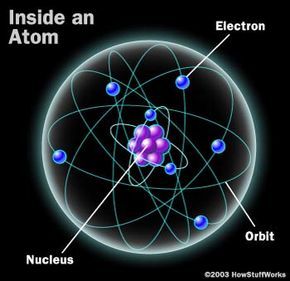 In its most basic representation, an atom comprises a nucleus surrounded by electrons in orbit.
HSW
In its most basic representation, an atom comprises a nucleus surrounded by electrons in orbit.
HSWThe universe is composed of roughly 100 distinct types of atoms. All matter we observe is formed through countless combinations of these atoms. The arrangement and bonding of these atoms determine whether they create water, metal, or even the carbonation in your soda.
Atoms are perpetually active, vibrating, rotating, and moving. Even the atoms in solid objects, like chairs, are in constant motion. Atoms can exist in various states of excitation, meaning they possess different energy levels. When sufficient energy is applied—through heat, light, or electricity—an atom can transition from its ground state to an excited state, depending on the energy input.
The image above provides a traditional depiction of an atom's structure.
This basic atomic model includes a nucleus (housing protons and neutrons) surrounded by an electron cloud. Electrons within this cloud orbit the nucleus in various paths, creating a dynamic system.
Absorbing Energy
 Energy absorption occurs when an atom takes in energy from sources like heat, light, or electricity. This can cause electrons to shift from a lower-energy orbit to a higher-energy one.
Energy absorption occurs when an atom takes in energy from sources like heat, light, or electricity. This can cause electrons to shift from a lower-energy orbit to a higher-energy one.Referring to the earlier illustration, while modern atomic models no longer show electrons in fixed orbits, visualizing these orbits as distinct energy levels helps in understanding atomic behavior.
In simpler terms, applying heat to an atom can cause electrons in lower-energy orbitals to jump to higher-energy ones farther from the nucleus. While this is a basic representation, it captures the fundamental principle behind how atoms function in laser technology.
When an electron transitions back to its ground state from a higher-energy orbit, it releases energy in the form of a photon, which is a particle of light.
Atoms releasing energy as photons is a common phenomenon. For instance, the red glow of a heated toaster element results from atoms emitting red photons when excited by heat. Similarly, the images on a TV screen are produced by phosphor atoms releasing light of various colors after being energized by high-speed electrons.
Light generation in devices like fluorescent lights, gas lanterns, and incandescent bulbs occurs through electrons shifting orbits and emitting photons.
The Laser-Atom Connection

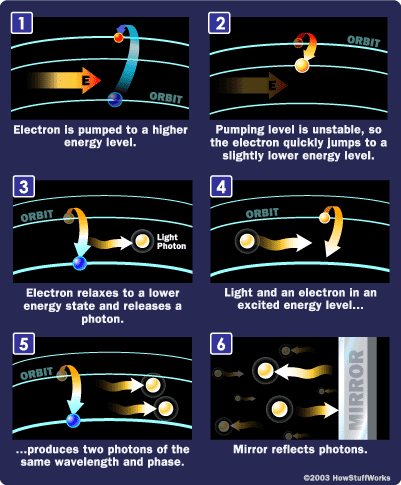
A laser is a mechanism that regulates how energized atoms emit photons. The term "laser" stands for light amplification by stimulated emission of radiation, which precisely explains its function.
While lasers come in various forms, they all share key characteristics. The lasing medium is energized to excite atoms, often using intense light flashes or electrical discharges, resulting in a high number of excited-state atoms (with higher-energy electrons). A substantial population of excited atoms is crucial for efficient laser operation.
Typically, atoms are excited to energy levels two or three steps above the ground state, enhancing the population inversion. Population inversion refers to the ratio of excited-state atoms to those in the ground state.
After the lasing medium is energized, it holds a group of atoms with electrons in excited states. These excited electrons possess higher energy levels than their relaxed counterparts. Just as energy was absorbed to reach this state, the electrons can release this energy by relaxing, emitting photons (light energy) in the process.
The emitted photon has a precise wavelength (color), determined by the electron's energy state at the time of release. Identical atoms with electrons in the same state will produce photons with matching wavelengths.
Laser Light
Laser light possesses distinct characteristics:
- The emitted light is monochromatic, consisting of a single wavelength (one specific color). This wavelength depends on the energy released as an electron transitions to a lower orbit.
- The light is coherent, meaning it is “organized” — all photons move in sync, with their wave fronts aligned in unison.
- The light is highly directional, forming a narrow, intense beam. In contrast, a flashlight emits light in multiple directions, resulting in a weaker and more dispersed output.
Stimulated Emission
Achieving these three properties requires a process known as stimulated emission. Unlike a standard flashlight, where atoms emit photons randomly, stimulated emission organizes photon release in a coordinated manner.
The wavelength of the photon emitted by an atom depends on the energy gap between its excited state and ground state.
When a photon with specific energy and phase interacts with another atom in the same excited state, stimulated emission can occur. The initial photon triggers the second atom to emit a photon that matches its frequency and direction.
Mirrors
A crucial component of a laser is a pair of mirrors positioned at either end of the lasing medium.
Photons with precise wavelengths and phases bounce between the mirrors, traveling through the lasing medium. This stimulates additional electrons to transition to lower energy levels, releasing more photons with matching wavelengths and phases.
This creates a cascade effect, rapidly generating numerous photons with identical wavelengths and phases. One mirror is partially reflective, allowing some light to pass through, which becomes the laser beam.
The next section's figures demonstrate these components, showing the workings of a basic ruby laser.
Ruby Lasers
A ruby laser includes a flash tube (similar to those used in cameras), a ruby rod, and two mirrors (one partially reflective). The ruby rod acts as the lasing medium, energized by the flash tube.
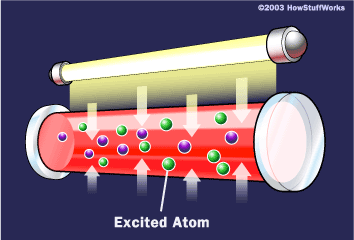 1. The flash tube activates, sending light into the ruby rod. This light energizes the atoms within the ruby.
Mytour
1. The flash tube activates, sending light into the ruby rod. This light energizes the atoms within the ruby.
Mytour 2. Some of these energized atoms release photons.
Mytour
2. Some of these energized atoms release photons.
Mytour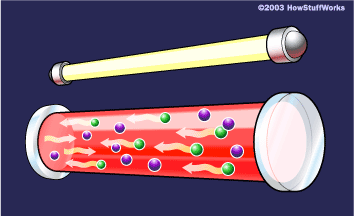 3. Certain photons travel parallel to the ruby's axis, reflecting between the mirrors. As they move through the crystal, they trigger additional atomic emissions.
Mytour
3. Certain photons travel parallel to the ruby's axis, reflecting between the mirrors. As they move through the crystal, they trigger additional atomic emissions.
Mytour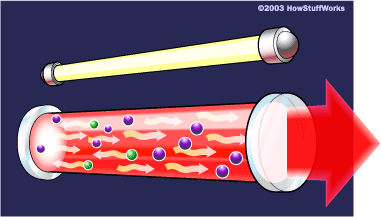 4. A coherent, single-wavelength, and collimated beam exits the ruby via the partially reflective mirror, producing laser light.
Mytour
4. A coherent, single-wavelength, and collimated beam exits the ruby via the partially reflective mirror, producing laser light.
MytourThree-Level Laser
This is how a three-level laser operates in real-world scenarios.
 Mytour
MytourThe following section will explore the various kinds of lasers.
Types of Lasers
Lasers come in various forms, with the lasing medium being either solid, gas, liquid, or semiconductor. They are typically categorized based on the material used for lasing.
Solid-state Lasers
In solid-state lasers, the lasing material is embedded within a solid matrix, such as in ruby or neodymium:yttrium-aluminum garnet (Yag) lasers. The neodymium-Yag laser produces infrared light at a wavelength of 1,064 nanometers (nm), where a nanometer equals 1x10-9 meters.
Gas Lasers
Helium and helium-neon (HeNe) lasers are among the most widely used gas lasers, primarily emitting visible red light. CO2 lasers, on the other hand, generate far-infrared energy and are ideal for cutting through tough materials.
Excimer Lasers
These lasers, named from 'excited' and 'dimers,' utilize reactive gases like chlorine and fluorine combined with inert gases such as argon, krypton, or xenon. Upon electrical stimulation, they form a pseudo molecule (dimer), which emits ultraviolet light when lased.
Dye Lasers
Dye lasers employ intricate organic dyes, such as rhodamine 6G, in liquid solutions or suspensions as their lasing medium. They offer the advantage of being adjustable across a wide spectrum of wavelengths.
Semiconductor Lasers
Often referred to as diode lasers, these are not classified as solid-state lasers. These compact electronic devices typically consume minimal power and can be integrated into larger systems, such as the writing mechanisms in certain laser printers or CD players.
What's Your Wavelength?
The ruby laser, illustrated earlier, is a solid-state laser that operates at a wavelength of 694 nm. Different lasing materials can be chosen depending on the required wavelength, power output, and pulse duration, as outlined in the table below.
Certain lasers, like the CO2 laser, are extremely powerful and capable of cutting through steel. The CO2 laser is particularly hazardous because it emits light in the infrared and microwave spectrum. Infrared radiation generates heat, allowing the laser to melt through materials it targets.
On the other end of the spectrum, diode lasers are relatively weak and commonly found in handheld laser pointers. These lasers usually produce a red beam with a wavelength ranging between 630 nm and 680 nm.
Lasers are widely used in industrial and research settings for various purposes, such as employing high-intensity laser beams to stimulate other molecules and analyze their reactions.
Below is a list of common lasers along with their respective emission wavelengths (in nanometers):
- Argon fluoride (UV): 193
- Krypton fluoride (UV): 248
- Xenon chloride (UV): 308
- Nitrogen (UV): 337
- Argon (blue): 488
- Argon (green): 514
- Helium neon (green): 543
- Helium neon (red) 633
- Rhodamine 6G dye (tunable): 570-650
- Ruby (CrAIO3) (red): 694
- Nd:Yag (NIR): 1,064
- Carbon dioxide (FIR): 10,600
Laser Classifications
Lasers are categorized into four main groups based on their potential to cause biological damage. Every laser should be marked with one of these four class labels for safety identification.
- Class I: These lasers are incapable of emitting radiation at levels considered hazardous.
- Class I.A.: This classification applies exclusively to lasers not meant for viewing, like those in supermarket scanners. Their power limit is capped at 4.0 mW.
- Class II: These visible lasers operate at low power, emitting above Class I levels but not exceeding 1 mW. The human eye's natural reaction to bright light provides protection.
- Class IIIA: These intermediate-power lasers (1-5 mW in continuous wave) pose risks only when viewed directly. Most laser pointers fall into this category.
- Class IIIB: These lasers are of moderate power.
- Class IV: High-power lasers (500 mW continuous wave or 10 J/cm pulsed) are dangerous under all viewing conditions, including scattered light. They also pose fire and skin hazards, requiring strict safety measures in facilities.
How Does a Laser Pointer Work?
Laser pointers function based on light amplification and stimulated emission. Within a laser diode, atoms or molecules are excited to release photons, creating a concentrated and coherent beam of light. This process, known as stimulated emission, ensures the photons are synchronized, producing the characteristic laser beam.
It’s crucial to recognize that laser pointers also emit infrared light, which is invisible to humans. While filters are used to block most of this light, responsible handling is essential to avoid directing the beam at reflective surfaces or eyes.
Laser light is monochromatic, consisting of a single wavelength, and coherent, with all photons moving in unison. This coherence allows the beam to remain tightly focused over long distances, minimizing divergence.
