 Water is the fundamental building block of life. Every living organism on Earth depends on it for survival. Trout55/Getty Images
Water is the fundamental building block of life. Every living organism on Earth depends on it for survival. Trout55/Getty ImagesIn its pure state, water has no smell, is almost clear, and lacks flavor. It exists within your body, the food you consume, and the drinks you enjoy. It’s used for cleaning your body, clothes, dishes, car, and nearly everything around you. You can sail on it or dive into it to escape the summer heat. Countless everyday products either contain it or were produced with its help. Every living being requires it, and without sufficient amounts, survival is impossible. It has been the center of political conflicts. In some regions, it’s a precious resource, hard to obtain, while in others, it’s readily available and often wasted. What is more vital to our survival than anything else? Water.
Fundamentally, water is a molecule composed of one oxygen atom and two hydrogen atoms, held together by shared electrons. Its V-shaped polar molecule structure gives it a positive charge near the hydrogen atoms and a negative charge near the oxygen atom. This polarity causes water molecules to attract and bond with each other, forming a hydrogen bond. This bond is responsible for many of water’s unique characteristics, including its higher density as a liquid compared to its solid form (ice floats on water). We’ll explore these fascinating properties in more detail later.
Water is unique as it naturally exists in three states: solid (ice), liquid, and gas (water vapor). It blankets roughly 70 percent of the Earth, totaling around 332.5 million cubic miles (1,386 million cubic kilometers) [source: U.S. Geological Survey]. The famous lines "Water, water, everywhere, nor any drop to drink" from the poem "The Rime of the Ancient Mariner" highlight that 97 percent of Earth's water is undrinkable due to its salt content (see illustration on next page). Only 3 percent is freshwater, with 77 percent of that locked in ice. Of the remaining 23 percent, a mere half a percent is accessible to sustain all plants, animals, and humans on the planet [source: National Geographic].
Is water really as simple as it seems? Not quite. Scientists are still uncovering many mysteries about it. Ensuring an adequate supply of clean, drinkable water for all living beings is a complex challenge. In this article, we’ll examine these issues, explore how plants, animals, and humans utilize water, and delve into what makes water so extraordinary.
The World's Water Supply
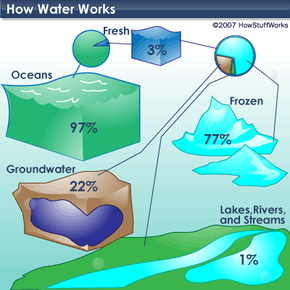 What is the source of our water?
What is the source of our water?News reports often discuss the world's shrinking water supply, but this isn’t entirely correct. The total amount of water remains constant, but the demand for it is rising rapidly. Some experts predict that the global population, currently at 6 billion, will double by 2050 [source: Cossi]. Additionally, pollution is reducing the amount of clean, drinkable water available.
In industrialized nations, accessing water is as simple as opening a tap, and it comes at a low cost. However, freshwater distribution is highly uneven globally. Over half of the world's water resources are concentrated in just nine nations: the United States, Canada, Colombia, Brazil, the Democratic Republic of Congo, Russia, India, China, and Indonesia [source: World Business Council for Sustainable Development]. While urban areas require vast amounts of water for purposes beyond drinking and sanitation, overpopulation in developing regions leaves many without even the most basic access.
The majority of the planet's freshwater — approximately 2.4 million cubic miles (10 million cubic kilometers) — is stored in underground aquifers. The remaining supply originates from:
- Rainfall (after accounting for evaporation): 28,500 cubic miles (119,000 cubic kilometers)
- Man-made reservoirs: 1,200 cubic miles (5,000 cubic km)
- Lakes: 21,830 cubic miles (91,000 cubic km)
- Rivers: 509 cubic miles (2,120 cubic km)
[source: World Business Council for Sustainable Development]
Water allocation is deeply tied to political borders, economic progress, and wealth disparities. For instance, in Mexico City, 9 percent of residents consume 75 percent of the available water, while aging infrastructure results in up to half of the supply being lost due to leaky pipes and evaporation [source: Cossi].
Many nations struggle to provide clean water for their rapidly expanding populations, lacking the financial means to build the infrastructure needed for purification and distribution. For instance, urban areas in China face severe water shortages, with much of the country's groundwater, lakes, and rivers contaminated. Approximately 700 million Chinese citizens rely on drinking water that fails to meet the standards set by the World Health Organization [source: WHO].
Middle Eastern countries have the lowest per capita water usage due to scarce natural freshwater sources. Conversely, the United States leads globally in water consumption, with each person using about 60,000 cubic feet (1,700 cubic meters) of water in 2002 [source: Organisation for Economic Co-operation and Development]. However, even within the U.S., certain states and regions face water scarcity. Coastal areas in Florida, for example, rely on freshwater piped from inland regions due to high saltwater levels, sparking political conflicts over water control.
Water Regulation
 This aerial image highlights the devastation of the Aral Sea caused by water diversion projects.
Photo courtesy NASA
This aerial image highlights the devastation of the Aral Sea caused by water diversion projects.
Photo courtesy NASAWater distribution and regulation are often managed by governments. In the United States, the Safe Drinking Water Act oversees this process. However, government control doesn't always benefit everyone. During the 1930s, the Soviet government diverted rivers feeding the Aral Sea (located between Kazakhstan and Uzbekistan) to irrigate cotton fields. This caused the sea's surface area to shrink by over 50 percent and its volume by 80 percent in the last 50 years [source: Swanson]. The sea's salinity rose, and it became contaminated with pesticides, fertilizer runoff, and industrial waste. The collapse of the fishing industry plunged the region into poverty, and pollutants from the exposed seabed have even been detected in the blood of Antarctic penguins [source: Swanson].
In some areas, water distribution has been privatized, frequently sparking disputes. During the late 1980s, the United Kingdom privatized its water boards (government-run water supply entities), which enhanced infrastructure. However, many were angered by the idea of companies profiting from such a fundamental necessity, particularly when those unable to pay faced severe consequences. This issue was later addressed through legislative measures.
In 2000 and 2005, Bolivia witnessed widespread protests against the privatization of its water supply. After foreign corporations assumed control of Bolivia's water systems, prices soared, making water unaffordable for the poor. In El Alto, "the expense of securing a water and sewage connection surpassed half a year's income at minimum wage" [source: Shultz]. The 2000 uprising, known as the "Bolivian Water Wars," resulted in martial law and over 100 injuries. Following these events, the Bolivian government terminated the private contracts.
Today, over a billion people, roughly 17 percent of the global population, lack access to clean water [source: World Health Organization]. Numerous governmental and nongovernmental organizations, such as UNICEF and Water Aid, are working to provide sustainable drinking water and sanitation solutions to impoverished communities in Asia and Africa. Water scarcity also affects the United States, where many states have initiatives to help disadvantaged individuals access sufficient water and manage utility bills.
Clearly, water scarcity is a significant issue. But what makes it so critical? In the following section, we’ll explore the vital role water plays in the human body.
Reducing water demand can be achieved through year-round conservation efforts. While many regions impose restrictions on lawn and garden watering during shortages, here are simple steps you can take independently:
- Turn off the tap while brushing your teeth or shaving.
- Opt for shorter showers.
- Only run your dishwasher and washing machine with full loads.
- Store drinking water in the fridge instead of running the tap.
- Choose a car wash that recycles water.
For additional advice, visit this link from the EPA.
Human Water Consumption
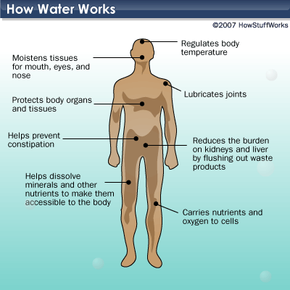 Why is water essential for humans?
Why is water essential for humans?Approximately 60 percent of our bodies consist of water [source: Mayo Clinic]. Water helps regulate body temperature, transports nutrients to cells, maintains moisture in mucous membranes, and removes waste. Our lungs are 90 percent water, our brains are 70 percent water, and our blood is over 80 percent water. In short, we cannot survive without it. On average, people lose about two cups of water daily (0.5 liters) through sweat. We also exhale a little more than a cup (237 ml) and expel around six cups (1.4 liters) each day. Additionally, we lose electrolytes — minerals like sodium and potassium that balance bodily fluids. So, how do we replenish it?
Around 20 percent of our daily water intake comes from the food we eat. Certain foods, such as watermelon, are almost entirely water. While daily water needs vary, the general recommendation is about eight cups (2 liters). Instead of obsessing over this amount, simply drink when thirsty. Beverages other than water can contribute to hydration, but some, like alcohol, may increase dehydration.
Dark yellow urine may indicate insufficient water intake. Naturally, you require more water during exercise, illness (such as diarrhea, vomiting, or fever), or prolonged exposure to hot climates. While most people can survive only a few days without water, this varies based on health and environmental conditions. Some individuals have reportedly lasted up to two weeks. Followers of a Buddhist boy meditating in Nepal claim he has gone two years without food or water, though doctors have not verified this [source: All Headline News].
Inadequate water intake or excessive water loss leads to dehydration. Symptoms of mild dehydration include dry mouth, intense thirst, dizziness, lightheadedness, and fatigue. Without proper hydration, severe dehydration can occur, resulting in convulsions, rapid breathing, a weak pulse, sagging skin, and sunken eyes. In extreme cases, dehydration can cause heart failure and death.
In developing nations, dehydration from diarrhea is a leading cause of mortality. Nearly 2 million people, primarily children, die annually from this condition [source: WHO]. Drinking water contaminated with biological agents and lacking proper sanitation can lead to diseases such as malaria, cholera, and infections from parasites like cryptosporidiosis and schistosomiasis. Water can also be tainted with chemicals, pesticides, and natural toxins.
On the following page, we’ll explore methods for purifying water.
Water Purification
Water deemed safe for consumption is referred to as potable water or drinking water, as opposed to safe water, which is suitable for bathing or cleaning. In the United States, the Environmental Protection Agency establishes maximum allowable levels for the 90 most common contaminants. If your water supply is compromised, your provider must notify you and advise on necessary precautions.
Water purification involves six fundamental steps.
- During coagulation, substances like lime and alum are introduced to the water, causing particles to cluster together.
- Next, the water is agitated to form larger clusters, known as flocs.
- The sedimentation process requires the water to sit for 24 hours, allowing the clusters to settle at the bottom.
- The water is then filtered, disinfected (typically with chlorine), and aerated.
- Aeration helps eliminate specific contaminants such as radon.
In the next section, we’ll examine how water circulates within animal and plant cells in greater detail.
If your water supply becomes contaminated and bottled water is unavailable, there are several methods to purify it. For cloudy water, first filter it through clean cloth or let it settle, then pour off the clear portion. Boil the water for one minute to eliminate most harmful organisms.
Alternatively, add one-eighth teaspoon of household chlorine bleach per gallon of water (or follow label instructions). Double the amount if the water is cloudy or discolored. Stir and let it sit for 30 minutes. Chlorine bleach tablets, available at camping stores, can also purify water. Another option is to use five drops of iodine per gallon for disinfection.
Store boiled or treated water in clean, covered containers. If the boiled water tastes flat or the chlorine flavor is too strong, transfer it between containers to improve taste.
Plant and Animal Water Consumption
 Water helps regulate plant temperature and facilitates nutrient transport.
Martin Poole/Digital Vision/Getty Images
Water helps regulate plant temperature and facilitates nutrient transport.
Martin Poole/Digital Vision/Getty ImagesPlants contain a higher percentage of water than animals, typically ranging from 90 to 95 percent [source: BBC]. Similar to animals, water in plants regulates temperature and transports nutrients. However, instead of consuming water through eating and drinking, plants absorb it via dew, irrigation, and rainfall.
Plants absorb water through their roots, and green plants utilize it in photosynthesis, the process by which they produce sugar for energy. (For more on photosynthesis, see How the Earth Works.) Water also provides structural support to plants. The pressure from osmosis — the movement of water into plant cells — maintains the rigidity of cell walls.
When you water a plant, it absorbs the liquid through capillary action. The water then moves from the roots via xylem vessels to the leaves, where it exits through tiny openings called stomata. These stomata open when the plant needs to cool down, a process known as transpiration, akin to sweating in humans and some animals. Carbon dioxide also enters the plant through these stomata.
Processing water in animals and humans is more complex than in plants, though there are similarities. Water ingested is absorbed in the upper small intestine via osmosis, entering the bloodstream and circulating throughout the body. Unlike plant cells, animal cells lack cell walls, necessitating circulatory systems to prevent cells from absorbing excessive water and salt, which could cause swelling. Our circulatory systems distribute water and eliminate excess through sweating and urination.
Certain animals, like the microscopic tardigrade, can survive without water for extended periods. In water-scarce environments, the tardigrade enters a state called anhydrobiosis, where sugar replaces water in its cells, making it resistant to extreme temperatures. Its metabolism slows, allowing it to remain in this near-lifeless state until water becomes available again.
Some plants have evolved unique adaptations to thrive with minimal water. One such adaptation is Crassulacean Acid Metabolism (CAM) photosynthesis. In CAM photosynthesis, plants store carbon dioxide as acid and keep their stomata closed during the day to reduce water loss (evaporation is slower at night). In extremely dry conditions, they may keep their stomata closed entirely. Cacti, for example, use CAM photosynthesis to endure the intense heat and drought of deserts.
Next, we’ll explore how the hydrologic, or water, cycle operates.
The Water Cycle
 Water vapor in clouds eventually condenses into droplets and falls as rain, sleet, hail, or snow.
Pete Turner/The Image Bank/Getty Images
Water vapor in clouds eventually condenses into droplets and falls as rain, sleet, hail, or snow.
Pete Turner/The Image Bank/Getty ImagesThe water cycle describes the endless movement of water on, above, and below the Earth. As noted earlier, water never disappears—it simply changes form. The sun powers this cycle through two key processes: condensation and evaporation. When the sun heats water surfaces, it evaporates into the atmosphere as vapor. As it cools and rises, it forms clouds, which eventually condense into droplets. Depending on atmospheric conditions, this water precipitates as rain, sleet, hail, or snow.
Some precipitation is intercepted by tree canopies and evaporates back into the air. Precipitation reaching the ground becomes runoff, which can form snow caps, glaciers, or seep into the soil to replenish aquifers. Aquifers are vast underground reservoirs of groundwater that can be tapped for use. Runoff also originates from snowmelt, triggered by sunlight and climate changes. Eventually, some runoff returns to lakes and oceans, where the sun evaporates it again. For more on the water cycle, see How the Earth Works.
Water that soaks into the soil may evaporate and return to the atmosphere. However, groundwater, a primary source of drinking water, can accumulate in aquifers over millennia. Unconfined aquifers have the water table as their upper boundary, where water pressure matches atmospheric pressure. Confined aquifers, often located beneath unconfined ones, are capped by layers of rock or other materials.
In the U.S., the oldest groundwater, called fossil water, is stored in the Ogallala Aquifer. Spanning approximately 175,000 square miles (450,000 square kilometers) across eight Great Plains states, the Ogallala Aquifer holds about 2,900 million acre-feet (3,600 million cubic kilometers) of water [source: High Plains/Ogallala Aquifer]. Formed 2 to 6 million years ago during the rise of the Rocky Mountains, the aquifer’s water is being depleted faster than it can be replenished due to the region’s arid climate. This has led scientists to describe its use as water mining.
Groundwater might also be present on other planets. Images captured by the Mars Global Surveyor spacecraft suggest the presence of gullies formed by flowing water on Mars. According to NASA, this water likely lies 300 to 1,300 feet (100 to 400 meters) beneath the surface. Europa, a moon of Jupiter, may also harbor subsurface water. As Earth’s water demand surpasses its supply, scientists speculate about the possibility of extracting water from other planets and moons in our solar system.
Water possesses numerous unique and remarkable properties that make it essential for life. These properties drive our ongoing efforts to find better ways to access and conserve it. In the next section, we’ll explore these characteristics and delve deeper into the nature of water itself.
Water Properties
 A water strider showcases surface tension.
Steve Maslowski/Getty Images
A water strider showcases surface tension.
Steve Maslowski/Getty ImagesThe hydrogen bonds between water molecules, discussed earlier, give rise to two of water’s unique properties: cohesion and adhesion. Cohesion refers to water’s ability to stick to itself, while adhesion describes its ability to bond with other surfaces. This is why water forms thin films on materials like glass. When water interacts with such surfaces, adhesive forces overpower cohesive forces, causing it to spread out rather than clump together.
Water exhibits a high degree of surface tension. This occurs because surface molecules are not surrounded by others on all sides, so they are pulled inward by cohesive forces from deeper molecules. These molecules bond strongly to each other but weakly to other surfaces. For example, water forms beads on waxy surfaces like leaves or waxed cars. Surface tension shapes these droplets into spheres, minimizing their surface area.
Capillary action is another outcome of surface tension. As discussed, this phenomenon occurs in plants as they draw water upward. Water adheres to the inner walls of plant tubes, while surface tension tries to flatten it. This causes the water to rise and re-cohere, repeating the process until gravity counteracts the buildup.
Water’s hydrogen bonds also explain why its solid form, ice, floats on liquid water. Ice is less dense because water molecules form crystalline structures at freezing temperatures (32°F or 0°C). Water’s thermal properties are tied to these bonds as well. It has a high specific heat capacity, requiring 4.2 joules per gram to raise its temperature by one degree Celsius. Water also has a high heat of vaporization, meaning it can absorb significant heat without a substantial temperature increase. This property influences climate, as oceans take a long time to warm.
Water is often called the universal solvent because it dissolves many substances. Substances that dissolve in water are hydrophilic, meaning their cohesive forces match or exceed water’s. Polar substances like salt and sugar dissolve easily in water. Conversely, hydrophobic substances, such as oil, do not dissolve, leading to the saying "oil and water don't mix." Water’s solvency means it is rarely pure, often containing dissolved minerals.
The presence of minerals distinguishes hard water from soft water. Hard water typically contains calcium, magnesium, and sometimes metals. While soap doesn’t lather well in hard water, it is generally safe. However, it can cause lime scale buildup in pipes, water heaters, and toilets.
Recent debates about water’s properties focus on the behavior of ice as it melts. Some researchers argue that its structure remains largely unchanged from its solid state, with only some hydrogen bonds breaking. Others propose that melting ice forms a completely different structure. Despite water’s critical role in life, there is still much we don’t fully comprehend about it.
