 In Los Angeles, on March 27, 2008, paleontologists from the Natural History Museum of Los Angeles were engaged in the delicate task of preparing and assembling the fossils of a 66-million-year-old Tyrannosaurus rex, affectionately named Thomas. This T-Rex specimen, estimated to be 70% intact, is one of only 30 partially complete T-Rex fossils in the world. Excavation of Thomas took place in Montana between 2003 and 2005. GABRIEL BOUYS/AFP/Getty Images
In Los Angeles, on March 27, 2008, paleontologists from the Natural History Museum of Los Angeles were engaged in the delicate task of preparing and assembling the fossils of a 66-million-year-old Tyrannosaurus rex, affectionately named Thomas. This T-Rex specimen, estimated to be 70% intact, is one of only 30 partially complete T-Rex fossils in the world. Excavation of Thomas took place in Montana between 2003 and 2005. GABRIEL BOUYS/AFP/Getty ImagesImportant Highlights
- Carbon-14 dating works by comparing the carbon-12 to carbon-14 ratio in a sample to that in a living organism, which helps determine the age of once-living organisms with considerable accuracy.
- With a half-life of 5,700 years, carbon-14 is effective for dating organic materials up to around 60,000 years old, and a specific formula is used to calculate the conventional radiocarbon age (CRA) of a sample.
- Though carbon-14 is widely used for dating, other isotopes like potassium-40 and uranium-235 follow the same principle. However, the reliability of radioisotope dating may be affected for materials that died after the 1940s due to shifts in carbon ratios caused by nuclear testing and burning fossil fuels.
You may have come across captivating stories in the news about ancient relics. For instance, during an archaeological excavation, a piece of wood is discovered and determined to be 5,000 years old. Another story involves an archaeologist who uncovers a child mummy in the Andes, revealing that the child lived more than 2,000 years ago. But how do scientists determine the age of such objects or human remains? What techniques do they use, and how do these methods function?
Radiocarbon dating, often referred to as carbon dating, is a method used to estimate the age of certain archaeological artifacts of biological origin, dating back as far as 50,000 years. This technique is commonly applied to materials such as bones, fabric, wood, and plant fibers created by human activities relatively recently.
How Carbon-14 Is Created
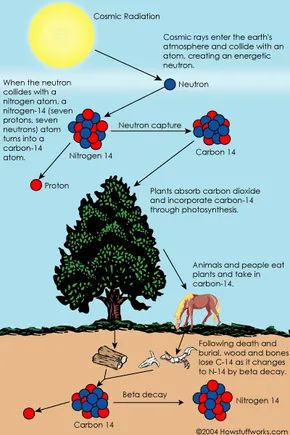
Every day, large quantities of cosmic rays strike the Earth’s atmosphere. For example, each person is bombarded by approximately half a million cosmic rays each hour. It’s not unusual for a cosmic ray to collide with an atom in the upper atmosphere, generating secondary cosmic rays in the form of energetic neutrons, which then collide with nitrogen atoms.
When a neutron strikes a nitrogen-14 atom (with seven protons and seven neutrons), it transforms into a carbon-14 atom (with six protons and eight neutrons) and a hydrogen atom (with one proton and no neutrons). Carbon-14 is a radioactive isotope, and its half-life is about 5,700 years.
For additional details about cosmic rays, half-life, and the process of radioactive decay, explore how nuclear radiation works.
Carbon-14 in Living Organisms
The carbon-14 atoms produced by cosmic rays combine with oxygen to form carbon dioxide. Plants absorb this carbon dioxide naturally and incorporate it into plant fibers through photosynthesis (carbon exchange). Animals and humans then consume plants, taking in carbon-14 as well.
The ratio of 'normal' carbon (carbon-12) to carbon-14 in the atmosphere and in living organisms remains nearly constant. Approximately one in a trillion carbon atoms are carbon-14. These carbon-14 atoms decay and are replaced by new carbon-14 atoms at a steady rate. Right now, your body contains a certain percentage of carbon-14 atoms, and this same percentage is present in all living plants and animals.
How the Radiocarbon Dating Technique is Applied
Once a living organism dies, it no longer takes in new carbon. The carbon-12 to carbon-14 ratio at the time of death is identical to that of any other living organism, but after death, the decaying carbon-14 is not replenished.
Carbon-14 decays according to its half-life of 5,700 years, while the amount of carbon-12 in the sample remains constant. By comparing the ratio of carbon-12 to carbon-14 in a sample with that in a living organism, it’s possible to estimate the age of the organism that once lived with remarkable accuracy.
The formula to calculate the conventional radiocarbon age (CRA) of a sample through carbon-14 dating is as follows:
where:
- ln represents the natural logarithm
- Nf/No is the ratio of carbon-14 in the sample compared to the amount present in living tissue
- t1/2 is the Libby half-life of carbon-14, which is 5,700 years
For instance, if you had a fossil that contained 10 percent of the carbon-14 found in a living organism, the calculation for the fossil’s age would be:
t = [ ln (0.10) / (-0.693) ] x 5,700 years
t = [ (-2.303) / (-0.693) ] x 5,700 years
t = [ 3.323 ] x 5,700 years
t = 18,940 years old
Due to carbon-14's half-life of 5,700 years, it is most effective for dating organic matter up to roughly 60,000 years old. However, the carbon-14 dating method is also applicable to other isotopes.
Potassium-40, another naturally occurring radioactive element in the human body, has a half-life of 1.3 billion years. Other important radioisotopes used for dating include uranium-235 (half-life = 704 million years), uranium-238 (half-life = 4.5 billion years), thorium-232 (half-life = 14 billion years), and rubidium-87 (half-life = 49 billion years). The use of multiple radioisotopes enables accurate dating of both biological and geological samples.
However, the accuracy of radioisotope dating might decline in the future. Anything that died after the 1940s, when nuclear tests, nuclear reactors, and the combustion of fossil fuels began to alter carbon ratios, will be more difficult to date with precision.
The Calibration Curve
Radiocarbon dating reveals the time since an organism's death, but it does not provide the exact calendar age. To find the calendar dates for organic materials, scientists calibrate their radiocarbon results by using known-age objects, like tree rings.
IntCal is an international collective that utilizes data from tree rings and ocean samples to create calibration curves. These curves are then applied by scientists to radiocarbon-dated materials to enhance the precision of their age estimates.
Calibration curves are created separately for different regions, such as the deep ocean, Southern Hemisphere, and Northern Hemisphere, in order to account for varying radiocarbon levels in each location.
