 The Molecule
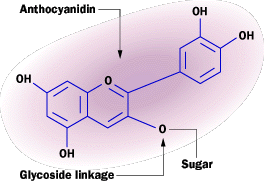
The MoleculeNature employs color in various ways. For many animals, color serves as camouflage. In contrast, plants often use color to attract insects for pollination, such as in flowers. The purple hue in red cabbage originates from a class of pigments called anthocyanins. These pigments are also present in flower petals, leaves (which turn red in autumn!), and fruits like blueberries. Anthocyanins are plant pigments categorized as flavonoids, responsible for the red, pink, violet, and magenta colors found in various plant parts.
The anthocyanidin group (See the sidebar below for the molecular structure) governs the pigment's color. The ring structures' electrons interact with incoming light, absorbing different frequencies. (For more on absorption, refer to How Light Works.) The molecule appears purplish-red because its electrons absorb the yellow, green, and blue portions of the visible spectrum.
The color of anthocyanins changes based on the surrounding acid or alkali level (i.e., pH). Because their color is influenced by the pH of the environment, these molecules can indicate the pH of any substance. When you create an extract from red cabbage juice, it shifts color when combined with solutions of different pH levels. In acidic solutions, anthocyanin turns bright pink, in neutral solutions it becomes reddish-purple, and in alkaline solutions, it turns green. This experiment demonstrates how to create a pH indicator using red cabbage!
Here are some fascinating resources:
- How to create a pH indicator using red cabbage
- Anthocyanin Biosynthesis
- The Changing Colors of Autumn
- Adding Color to Food through Juicing
- Understanding How Cells Function
