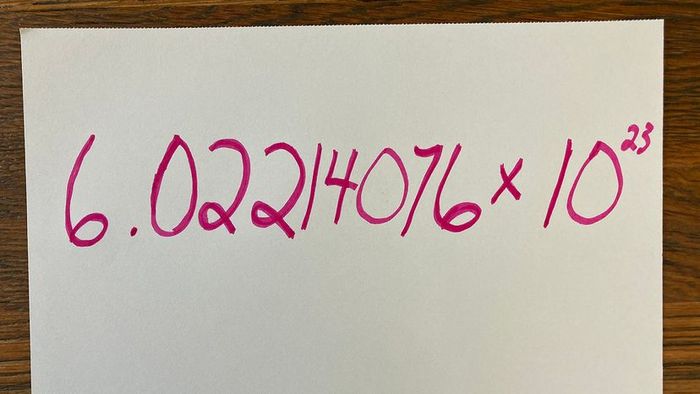 Avogadro's number: It's certainly easier to recall than pi. A. Loudermilk
Avogadro's number: It's certainly easier to recall than pi. A. LoudermilkHere’s a clue: It’s definitely not 867-5309. That’s Jenny's number, not Avogadro’s. You also won’t find these digits spray-painted on a bathroom wall. However, you can find it in any basic chemistry textbook: 6.02214076 × 10. If you prefer to visualize it, that's 602,214,076,000,000,000,000,000 [source: NIST]. In a rush? Just call it a mole.
Much like a dozen represents 12 items, a mole is simply Avogadro’s number of things. In the realm of chemistry, those ‘things’ are atoms or molecules. Sure, you could have a mole of baseballs or anything else, but considering that a mole of baseballs would stack high enough to cover the Earth several hundred miles up, you won’t find much practical use for a mole of anything larger than a molecule. So how did Amedeo Avogadro (full name: Lorenzo Romano Amedeo Carlo Avogadro) and the concept of chemistry intersect?
Born in Italy in 1776, Avogadro grew up during a pivotal era in chemistry's evolution. Chemists like John Dalton and Joseph Louis Gay-Lussac were making strides in understanding atoms and molecules, and they frequently debated how these incredibly tiny particles behaved. Avogadro was particularly intrigued by Gay-Lussac's law of combining volumes, which stated that when two volumes of gases react to form a third gas, the ratio of the volumes of reactants to product is always in simple whole numbers. For example: Two volumes of hydrogen gas combine with one volume of oxygen gas to produce two volumes of water vapor (provided temperatures are high enough), with no excess, as shown here:
2H2 + O2 --> 2H2O
By exploring the consequences of this law, Avogadro concluded that for the law to hold true, equal volumes of any two gases at the same temperature and pressure must contain the same number of particles (Avogadro's law). To explain this, he proposed that there must be a distinction between atoms and molecules and that some elements, like oxygen, exist as molecules (for oxygen, O2 instead of just O). Though Avogadro didn’t have the term 'molecule' at his disposal to describe his theory, and despite resistance from John Dalton and others, it was eventually Stanislao Cannizzaro who championed Avogadro’s ideas, bringing them the recognition they deserved. By that time, Avogadro had already passed away.
So, where does Avogadro's number fit into all of this? Because Avogadro's law played such a crucial role in the development of chemistry, chemist Jean Baptiste Perrin honored him by naming the number after him. Read on to discover how scientists determined Avogadro's number and why it remains a cornerstone of chemistry today.
Avogadro's Number in Action
How did chemists come up with such a seemingly random value for Avogadro's number? To understand its origin, we need to first dive into the concept of the atomic mass unit (amu). The atomic mass unit is defined as one-twelfth of the mass of a single carbon-12 atom (the most abundant isotope of carbon). Here’s why this is interesting: Carbon-12 consists of six protons, six electrons, and six neutrons, and since electrons have very little mass, one-twelfth of the mass of a carbon-12 atom is nearly identical to the mass of a single proton or neutron. The atomic weights of elements (those numbers under the elements on the periodic table) are also measured in atomic mass units. For example, hydrogen has an atomic weight of 1.00784 amu on average.
Unfortunately, chemists don’t have a scale that can directly measure atomic mass units, nor can they measure a single atom or molecule to perform a reaction. Since atoms vary in weight, chemists needed to find a way to bridge the gap between the microscopic world of atoms and the practical world of lab scales that measure in grams. To achieve this, they created a relationship between the atomic mass unit and the gram, which is expressed as follows:
1 amu = 1/6.02214076 x 10 grams
This relationship means that if we had Avogadro’s number, or one mole, of carbon-12 atoms (which, by definition, has an atomic weight of 12 amu), the weight of that sample of carbon-12 would be exactly 12 grams. Chemists use this connection to easily convert between the measurable unit of grams and the invisible unit of moles, atoms, or molecules.
Now that we understand the usefulness of Avogadro’s number, let’s address one final question: How did chemists determine how many atoms make up a mole in the first place? The initial rough estimate came from physicist Robert Millikan, who measured the charge of an electron. By the time Millikan made his breakthrough, the charge of a mole of electrons, known as a Faraday, had already been determined.
By dividing a Faraday by the charge of an electron, we arrive at Avogadro’s number. Over the years, scientists have developed increasingly precise methods for estimating this value, including advanced techniques such as using X-rays to analyze the geometry of a 1-kilogram silicon sphere and calculating the number of atoms it contained from that data. While the kilogram serves as the foundation for all mass units, some scientists propose using Avogadro’s number as the basis, much like how the length of a meter is currently defined by the speed of light rather than the traditional method.
While you probably won’t get the day off work or find greeting cards at your local store, Mole Day is celebrated annually by chemists worldwide. Since Avogadro’s number is 6.022 × 10, it only makes sense that the festivities begin at 6:02 a.m. on October 23rd. Participants enjoy chemistry-themed jokes, blow bubbles of natural gas and set them on fire, toast with drinks cooled by dry ice, and even recite the mole pledge of allegiance.
Special thanks to Meisa Salaita, chemistry expert and Director for Education & Outreach at the NSF Center for Chemical Evolution, for her help with this article.
