1. Combustion
In simple terms, combustion is a chemical reaction between a substance and oxygen that results in flames. Some of the most familiar examples of combustion occur in the kitchen (when cooking with gas, charcoal, or kerosene), in a romantic setting with a candle, or when lighting a cigarette with a match or lighter. Combustion refers to a high-temperature oxidation-reduction reaction between a fuel and an oxidizing agent, usually atmospheric oxygen, producing vaporized oxidation products that form smoke. Combustion produces flames and generates enough heat to sustain the reaction. Almost anything can burn, which makes fire safety extremely important.
Combustion can be detected by its characteristic smells: burning electrical wires, rubber, cotton fibers, or sweet scents like honey and sugar, along with sharp fumes such as SO2, SO3, and chlorine. Furthermore, combustion is always accompanied by smoke, which varies in color depending on the material burning and the oxygen availability. The reaction is also marked by visible flames and occasional popping sounds.
For instance, the combustion reaction of propane, commonly used in gas grills and some heaters, is as follows:
C3H8 + 5O2 → 4H2O + 3CO2 + energy.


2. Rusting
Over time, the outer surface of iron rods develops a flaky red layer known as rust. This is a classic example of an oxidation reaction. Everyday instances of rusting include the formation of verdigris (a greenish rust) on copper, brass, and bronze surfaces, and the tarnishing of silver.
Rust (or iron oxide) is the result of iron reacting with oxygen in the presence of water or moisture. The rust forms fragile, flaky layers, typically brown, reddish-brown, or red in color. These rust layers do not offer protection to the underlying iron. If given enough time, oxygen and water will completely corrode and decompose any iron object.
The cause of rusting is the interaction between iron or its alloys (like steel) with oxygen and moisture over a prolonged period, leading to the formation of iron oxide, commonly known as rust. The primary catalyst for this process is water. Although the iron or steel seems solid, water molecules can infiltrate tiny pores and cracks in the metal. The hydrogen atoms in the water combine with other elements to form acids, which corrode the iron, making it more exposed. In marine environments, corrosion can occur more rapidly. Meanwhile, oxygen atoms combine with iron atoms to form iron oxide, which weakens the metal and makes it brittle and porous. As a result, rusting accelerates the corrosion process, making maintenance critical.
Below is the chemical equation representing the rusting process of iron: Fe + O2 + H2O → Fe2O3 . XH2O


3. Combining Chemicals
In reality, many chemicals combine with each other in everyday life. These chemical reactions are easy to perform, yet they have numerous applications in households and daily routines. For instance, if you mix vinegar with baking soda, you create a small volcano, or combine milk with baking powder in a recipe, you get double the volume or produce a reaction that causes fizzing when you add other ingredients. These combined elements generate carbon dioxide gas and water. The carbon dioxide bubbles help the volcano eruption or make the cake rise as it bakes.
These simple chemical reactions in daily life bring surprising benefits. For example, when baking soda reacts with vinegar, it can serve multiple purposes. Baking soda, a leavening agent, contains cleaning properties that can remove stains and grime from objects, while vinegar is a strong acid, also effective in cleaning surfaces. When these two substances mix, they trigger a fizzing reaction that helps break down dirt and grime. The reaction works wonders to loosen stubborn dirt, making it easier to clean your belongings. In addition to this chemical reaction for cleaning, you can also use baking soda and vinegar individually for the same purpose on various items.
Here’s the overall chemical equation for the reaction between baking soda and vinegar: HC2H3O2 (aq) + NaHCO3 (aq) → NaC2H3O2 (aq) + H2O + CO2 (g)


4. Battery
In everyday life, there are many devices that require the use of batteries. Items like fans, clocks, and toys all rely on batteries. A battery, also referred to as a "battery" in English, transforms chemical energy into electrical energy. Since its invention by Alessandro Volta in 1800, the battery has become a common power source for household items and industrial applications.
Batteries operate using electrochemical reactions or redox reactions, which convert chemical energy into electrical power. These natural oxidation reactions occur in the cells of the battery, while artificial chemical reactions happen in electrochemical cells. In essence, a battery shares similarities with an accumulator, but a key difference is that a battery is designed to store energy temporarily and will cease to function once the chemical compounds are used up, requiring recharging from an external power source. On the other hand, a fuel cell does not store fuel internally, and instead, it continuously generates electricity as long as it is supplied with an external fuel source. Compared to conventional batteries, fuel cells produce little to no environmental pollution, and the water generated as a byproduct of the reaction is clean, drinkable water.


5. Digestion
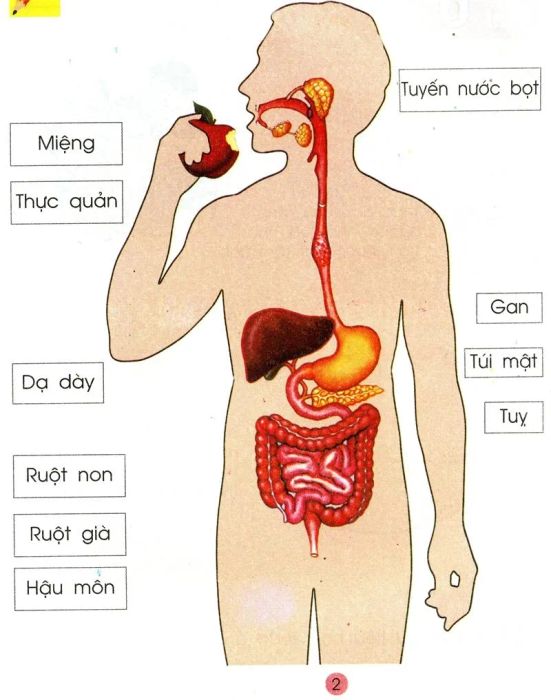
The digestive process is a vital activity that occurs daily in the bodies of all animals. Yet, in reality, many do not fully understand the concept of digestion or how it unfolds. Digestion is the process of transforming complex organic compounds into simpler nutrients that the body can absorb. It involves both mechanical and chemical changes in food, occurring in different organs throughout the body. In the end, the body absorbs the nutrients, and waste is eliminated.
Thousands of digestive processes take place every time we eat. As soon as food enters the mouth, an enzyme in saliva called amylase begins breaking down sugars and carbohydrates into simpler forms that the body can absorb. Then, when the food moves into the stomach, hydrochloric acid reacts with it to continue breaking it down. Simultaneously, enzymes break down proteins and fats, enabling their absorption into the bloodstream through the intestinal cells.

6. Acid-Base Reactions
Acids are substances containing one or more hydrogen atoms that, in solution, release positively charged hydrogen ions. An acid in aqueous solution has a sour taste, turns blue litmus paper red, reacts with certain metals (such as iron) to release hydrogen, reacts with bases to form salts, and promotes certain chemical reactions (acid catalysis). Bases, on the other hand, are bitter and turn red litmus paper blue. They react with acids to form salts and catalyze specific chemical reactions (base catalysis).
When we combine an acid (like vinegar or lemon) with a base (such as baking soda or soap), we are conducting an acid-base reaction. These are neutralization reactions, producing salt and water.
For example, a general chemical equation for an acid-base reaction that produces potassium chloride: HCl + KOH → KCl + H2O.


7. Reactions of Soap and Cleaning Agents
Since ancient times, humans have utilized natural cleaning agents such as ash, rice husks, soapberry, soap nut, lemon, and salt to clean objects. Today, with advancements in technology, scientists have developed a variety of chemical cleaners for daily use, such as soaps and detergents.
Soap is a cleaning agent used to remove stains, grease, and bacteria. It is made from sodium or potassium salts of fatty acids. Soaps can be found in bar, powder, or liquid forms. In the past, soap was produced by reacting fats with alkalis through a process known as saponification. The result was sodium or potassium salts of fatty acids, leading to the classification of soaps into hard soap (containing sodium) and soft soap (containing potassium). However, these types of soap have a drawback: they do not clean effectively in hard water as they form precipitates with calcium and magnesium ions, which can damage fabric. So, the chemical reactions that we use daily to clean are based on these principles.
Soaps work by removing dirt and allowing it to be washed away with water. Detergents operate through surfactant action, reducing the surface tension of water so that it can interact with oils, break them up, and wash away dirt and bacteria.


8. Photosynthesis
Photosynthesis is one of the most common chemical reactions that occur daily, playing a crucial role in the lives of humans, animals, and plants. Photosynthesis can be considered as the process by which plants, algae, and certain bacteria absorb sunlight energy to create organic compounds, which they use for themselves and as a food source for almost all living organisms on Earth. This chemical energy is stored in carbohydrate molecules like sugar, which are synthesized from carbon dioxide and water.
Most plants, algae, and bacteria perform photosynthesis. Photosynthesis helps maintain the concentration of oxygen in the atmosphere and provides all the organic compounds and most of the energy required for life on Earth.
All life on our planet depends on photosynthesis because the products of photosynthesis are the organic materials that serve as food for all living organisms, as well as raw materials for industries and medicines for humans. Photosynthesis also supplies the energy needed to sustain the activities of living organisms. Notably, when plants photosynthesize, they transform water and CO2 (carbon dioxide) into nutrients and oxygen gas. Oxygen is essential for human and animal life, which is why planting trees is crucial, especially in densely populated areas. Photosynthesis regulates the air by releasing oxygen and absorbing CO2 (helping to prevent the greenhouse effect and climate change).
The general equation for photosynthesis:
6 CO2 + 12 H2O →→ C6H12O6 + 6 O2 + 6 H2O
(as MT, chlorophyll)


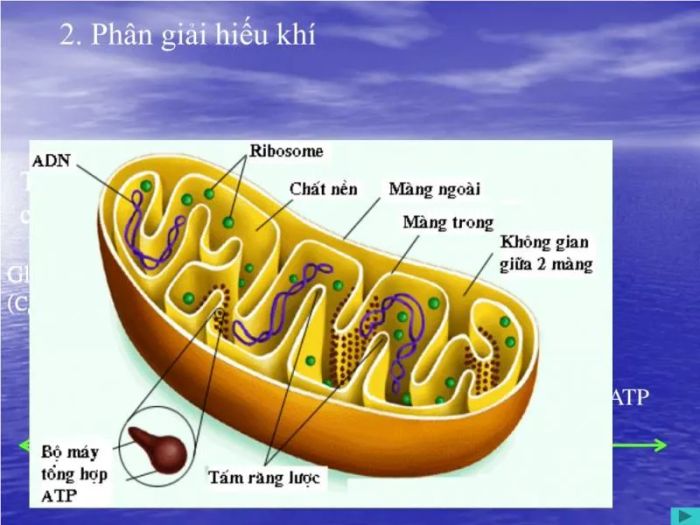
9. Aerobic Respiration
Aerobic respiration is a process that occurs in the presence of O2, requiring oxygen for the respiration to take place. Along with anaerobic respiration, aerobic respiration is a vital part of respiration in green plants.
Aerobic respiration is a process in which substrates are broken down to release energy necessary for sustaining the body's activities. The raw materials used are simple sugars, which undergo glycolysis and result in the final product of ATP. The most significant feature of aerobic respiration is that it requires an oxygen-rich environment to proceed.
Aerobic respiration holds crucial importance, as it helps synthesize ATP and provides the necessary materials for various synthetic reactions. The respiratory quotient (RQ) is the ratio between the number of CO2 molecules released and the number of O2 molecules absorbed during respiration. This ratio reveals the type of substances being respired and allows an evaluation of the plant's respiratory condition.
Chemical equation:
C6H12O6 + 6O2 → 6CO2 + 6H2O + Energy (36 ATPs).


10. Anaerobic Respiration
Anaerobic respiration, also known as anaerobic breathing, is the process by which carbohydrates are broken down to release energy for the cell. In this process, the final electron acceptor in the electron transport chain is an inorganic molecule other than molecular oxygen. For example, nitrate (NO3) serves as the final electron acceptor in nitrate respiration, while sulfate (SO4) is used in sulfate respiration.
These final electron acceptors have a lower reduction potential than O2, meaning they generate less energy for each oxidized molecule. As a result, anaerobic respiration is less efficient than aerobic respiration.
In anaerobic respiration, simple sugars are used, undergoing glycolysis to produce ATP. This process takes place in oxygen-deprived environments, specifically within the cytoplasm of eukaryotic organisms (lacking mitochondria). Anaerobic respiration is mainly employed by bacteria and archaea that live in oxygen-limited environments. Many anaerobic organisms are obligate anaerobes, meaning they can only respire using anaerobic compounds and will perish in the presence of oxygen.
Anaerobic respiration plays a crucial role in the global nitrogen, sulfur, iron, and carbon cycles by reducing the oxidation states of nitrogen, sulfur, and carbon into more reduced compounds. The biochemical cycling of these compounds, dependent on anaerobic respiration, has a significant impact on the carbon cycle and global warming.
Anaerobic respiration occurs in a variety of environments, including freshwater and marine sediments, soil, groundwater, deep underground environments, and biofilms. Even in oxygen-containing environments like soil, microenvironments exist where oxygen is absent due to the slow diffusion of oxygen.
One ecological example of the importance of anaerobic respiration is the use of nitrate as the final electron acceptor, or the reduction of dissolved nitrate, which is the primary pathway for returning fixed nitrogen to the atmosphere as molecular nitrogen. Another example is methanogenesis, a form of carbonate respiration, used to produce methane through anaerobic digestion. Biogas methane is a sustainable alternative to fossil fuels.
Chemical equation:
C6H12O6 → 2C2H5OH + 2CO2 + energy.