1. Pneumococcal Conjugate Vaccine (Prevenar 13)
Prevenar 13, also known as the pneumococcal 13-valent vaccine, is a new vaccine designed to prevent pneumonia, middle ear infections, meningitis, and sepsis caused by pneumococcal bacteria. Developed by Pfizer, a leading global pharmaceutical and biotechnology company, Prevenar 13 contains 13 different strains of the Streptococcus pneumoniae bacteria. This vaccine is intended for children from 6 weeks of age and older, as well as adults, particularly those who are beyond the age for receiving the Synflorix vaccine, or those with chronic conditions such as diabetes, chronic obstructive pulmonary disease (COPD), or compromised immune systems. Prevenar 13 works by stimulating the immune system to recognize the components of pneumococcal bacteria, allowing the body to produce antibodies more rapidly when exposed to the bacteria.
Contraindications:
- Do not administer Prevenar 13 to individuals who are allergic to any components of the vaccine or to diphtheria toxoid.
- Do not give Prevenar 13 to patients with thrombocytopenia or any bleeding disorders.
Vaccination Schedule for Children:
- First dose: Between 2 and 6 months of age.
- Second dose: At least 1 month after the first dose.
- Third dose: At least 1 month after the second dose.
 Pneumococcal Conjugate Vaccine (Prevenar 13)
Pneumococcal Conjugate Vaccine (Prevenar 13) Pneumococcal Conjugate Vaccine (Prevenar 13) for Children
Pneumococcal Conjugate Vaccine (Prevenar 13) for ChildrenMeasles is a highly contagious viral infection that spreads rapidly from person to person. It can cause severe complications such as pneumonia, middle ear infections, encephalitis (brain inflammation), mouth ulcers, corneal ulcers, and malnutrition. In pregnant women, it can lead to miscarriage or premature birth. Mumps is another infectious disease that can affect people of all ages. For men in their reproductive years, mumps can cause infertility. In pregnant women, mumps can harm the fetus, especially in the first trimester, leading to miscarriage. Rubella, similar to measles, is a dangerous viral infection that can cause miscarriage, stillbirth, or birth defects in a baby if the mother is infected during pregnancy. Due to the serious risks associated with measles, mumps, and rubella, the MMR combination vaccine is recommended for all children over 12 months of age, especially for women of childbearing age.
Contraindications:
- Individuals with a history of allergic reactions to neomycin.
- Those currently experiencing a fever or respiratory infection.
- Active tuberculosis that has not been treated, or individuals on immune-suppressing medications.
Vaccination schedule for children:
- First dose: Typically administered between 12 and 15 months of age, after the baby’s immune system has matured enough to avoid interference from maternal antibodies.
- Second dose: A booster shot given usually between the ages of 4 and 6, typically during preschool or kindergarten years.
 MMR Vaccine
MMR Vaccine MMR Vaccination for Children
MMR Vaccination for Children
Polio virus enters the human body through the digestive system and affects the central nervous system, causing damage to motor neurons. The virus spreads rapidly, making it a potential source of widespread outbreaks. The most vulnerable groups to polio infection include those who haven't received the polio vaccine. Pregnant women, the elderly, young children (especially those under 5 years old), and people with weakened immune systems are at the highest risk of infection. Polio has no cure and leaves many long-term disabilities. Treatment focuses on alleviating symptoms, possibly with physical therapy and portable ventilators for respiratory support. The only effective way to prevent polio is to actively build immunity through polio vaccination for children under 1 year old. This vaccine has been part of the Ministry of Health's expanded immunization program since 2010.
Contraindications:
- Children who had severe reactions after previous IPV or IPV-containing vaccine shots.
- Children allergic to any vaccine component or ingredients like neomycin, streptomycin, or polymyxin B.
- Children with severe organ dysfunction, such as respiratory failure, circulatory failure, heart failure, kidney failure, or acute liver failure, should delay vaccination until their health is stable.
Recommended Vaccination Schedule:
- 3 doses of oral polio vaccine (OPV) at 2, 3, and 4 months of age.
- At 5 months of age, children should receive 1 dose of the IPV vaccine.
 Polio Vaccine (IPV)
Polio Vaccine (IPV)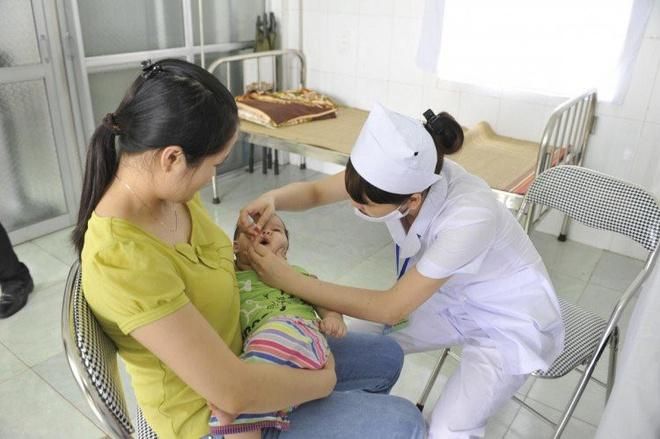 The IPV vaccine is designed to protect children from polio, a highly contagious virus that can cause permanent paralysis. This vaccine is administered through an injection and is a crucial part of early childhood immunization schedules. Ensuring your child receives the IPV vaccine on time is essential for their long-term health and well-being.
The IPV vaccine is designed to protect children from polio, a highly contagious virus that can cause permanent paralysis. This vaccine is administered through an injection and is a crucial part of early childhood immunization schedules. Ensuring your child receives the IPV vaccine on time is essential for their long-term health and well-being.The Rotavirus vaccine (RV) is an oral vaccine that protects against rotavirus infections, which can cause severe diarrhea in infants and young children. Rotavirus spreads easily through contaminated hands, surfaces, and bodily fluids, and is particularly dangerous in daycare settings or hospitals. Vaccination is the best way to prevent rotavirus-related illnesses, which can lead to dehydration, malnutrition, and even death if untreated. Infants under 5 years old, especially in cold and wet seasons, are most at risk. Rotavirus infections typically have a 2-3 day incubation period, with symptoms lasting 5-7 days, including vomiting, diarrhea, fever, and dehydration.
The Rotavirus vaccine schedule includes three doses: the first dose is given between 7.5 and 12 weeks of age, with subsequent doses administered at least 4 weeks apart. It is important to complete the vaccination schedule before the child reaches 32 weeks of age to ensure full protection against this potentially life-threatening virus.
 The oral Rotavirus vaccine is an essential immunization for young children, offering protection against severe diarrhea caused by rotavirus. This vaccine helps prevent dehydration and other complications associated with the illness, making it a key part of the childhood vaccination program.
The oral Rotavirus vaccine is an essential immunization for young children, offering protection against severe diarrhea caused by rotavirus. This vaccine helps prevent dehydration and other complications associated with the illness, making it a key part of the childhood vaccination program. Before administering the Rotavirus vaccine (RV) to your child, it's important to ensure they are in good health. Check for any signs of illness or previous reactions to vaccines. If your child is sick or has any medical conditions that may interfere with vaccination, consult with your pediatrician before proceeding with the vaccine.
Before administering the Rotavirus vaccine (RV) to your child, it's important to ensure they are in good health. Check for any signs of illness or previous reactions to vaccines. If your child is sick or has any medical conditions that may interfere with vaccination, consult with your pediatrician before proceeding with the vaccine.Pneumococcal bacteria are a leading cause of meningitis, particularly affecting children under 6 years old and the elderly. Meningitis caused by these bacteria can lead to severe complications, including blindness, deafness, paralysis, memory loss, and neurological disorders. The Synflorix vaccine provides protection against 10 of the most common pneumococcal strains, which are responsible for diseases like meningitis, pneumonia, and sepsis.
The Synflorix vaccine is contraindicated for individuals who are allergic to any of its components. It is essential to confirm that your child is not allergic to any ingredients before proceeding with vaccination.
 The vaccination schedule for pneumococcal meningitis includes three primary doses: the first at 2 months, the second at 3 months, and the third at 4 months of age. A booster dose is recommended 6 months after the third dose to maintain immunity against pneumococcal diseases.
The vaccination schedule for pneumococcal meningitis includes three primary doses: the first at 2 months, the second at 3 months, and the third at 4 months of age. A booster dose is recommended 6 months after the third dose to maintain immunity against pneumococcal diseases. The pneumococcal vaccine is essential for protecting against infections caused by the Streptococcus pneumoniae bacteria, which can lead to severe illnesses like meningitis, pneumonia, and blood infections. This vaccine is particularly important for children under 6 years old and older adults, who are at higher risk of complications. Synflorix, a vaccine covering 10 common pneumococcal strains, is an effective solution to prevent life-threatening diseases and long-term disabilities caused by these bacteria.
The pneumococcal vaccine is essential for protecting against infections caused by the Streptococcus pneumoniae bacteria, which can lead to severe illnesses like meningitis, pneumonia, and blood infections. This vaccine is particularly important for children under 6 years old and older adults, who are at higher risk of complications. Synflorix, a vaccine covering 10 common pneumococcal strains, is an effective solution to prevent life-threatening diseases and long-term disabilities caused by these bacteria.The flu is a highly contagious respiratory illness caused by influenza viruses, types A, B, and C. It spreads primarily through droplets from coughs and sneezes, and can also be transmitted by direct contact with contaminated surfaces. In some cases, humans can contract the flu from infected animals, such as poultry. The virus specifically targets respiratory epithelial cells, replicating rapidly and causing damage when it enters the bloodstream and other organs.
Children under 6 months old are too young to receive the flu vaccine. Additionally, individuals who have severe, life-threatening allergies to any components of the flu vaccine, such as gelatin or antibiotics, should avoid vaccination. Ensuring that the vaccine is safe for each individual is a crucial part of the immunization process.
 The flu vaccine is administered in two doses, spaced at least one month apart, to ensure full protection. Following the initial doses, a yearly booster shot is recommended to maintain immunity against circulating flu strains. This vaccination is key to preventing flu-related complications, especially in vulnerable populations such as young children, the elderly, and those with weakened immune systems.
The flu vaccine is administered in two doses, spaced at least one month apart, to ensure full protection. Following the initial doses, a yearly booster shot is recommended to maintain immunity against circulating flu strains. This vaccination is key to preventing flu-related complications, especially in vulnerable populations such as young children, the elderly, and those with weakened immune systems. Vacxin ngăn ngừa bệnh cúm
Vacxin ngăn ngừa bệnh cúm
7. Vacxin phòng ngừa viêm gan A
Viêm gan A là tình trạng nhiễm trùng ở gan do virus viêm gan A gây ra. Virus viêm gan A thường lây nhiễm qua việc sử dụng các loại thức ăn, nước uống đã bị nhiễm virus do vệ sinh kém. Ngoài ra, loại virus này còn có thể lây nhiễm qua việc tiếp xúc trực tiếp với người nhiễm bệnh. Người bị nhiễm virus viêm gan A có thể phát tán nguồn bệnh trong khoảng 2 tuần trước khi xuất hiện triệu chứng và ít nhất 1 tuần sau khi có triệu chứng điển hình. Các triệu chứng thường xuất hiện từ 15 - 50 ngày sau khi nhiễm virus viêm gan A. Triệu chứng thường gặp ở bệnh nhân viêm gan A gồm sốt khởi phát đột ngột, mệt mỏi toàn thân, giảm vị giác, buồn nôn và nôn ói, vàng da, vàng mắt, nước tiểu đậm màu, phân bạc màu... Nhiễm virus viêm gan A thường không dẫn đến bệnh lý gan mạn tính và hầu hết bệnh nhân có thể phục hồi trong khoảng 1 tháng. Để phòng ngừa viêm gan A, cần tránh sử dụng nguồn thức ăn và nguồn nước bị nhiễm bẩn, thực hiện ăn chín uống sôi. Đồng thời, tiêm vắc-xin phòng viêm gan A là biện pháp phòng bệnh hữu hiệu nhất.
Chống chỉ định:
- Người mẫn cảm với bất kỳ thành phần nào của vắc-xin. Đặc biệt những trường hợp có biểu hiện mẫn cảm với vắc-xin phòng viêm gan A ở lần tiêm trước
- Người mắc các bệnh bẩm sinh, mắc bệnh tim, bệnh thận, bệnh gan hay bị bệnh đái tháo đường hoặc suy dinh dưỡng, bệnh cấp tính... không phải là đối tượng chống chỉ định tiêm vắc-xin phòng viêm gan A...
Lộ trình tiêm cho bé:
- Liều đầu tiên khi trẻ đủ 12 tháng tuổi trở lên (đối với vắc-xin Avaxim 80 UI/0.5ml hoặc vắc-xin phòng viêm gan A và B: Twinrix 1ml) hoặc đủ 24 tháng tuổi trở lên (đối với vắc-xin Havax 0.5ml).
- Liều thứ hai cách liều thứ nhất 6 tháng.
 Vacxin phòng ngừa viêm gan A
Vacxin phòng ngừa viêm gan A The Hepatitis A vaccine is essential for preventing the transmission of the Hepatitis A virus, which can lead to severe liver damage. It is especially important for children, travelers to areas with high infection rates, and individuals at higher risk of contracting the virus. Vaccination ensures long-term protection and reduces the spread of the disease.
The Hepatitis A vaccine is essential for preventing the transmission of the Hepatitis A virus, which can lead to severe liver damage. It is especially important for children, travelers to areas with high infection rates, and individuals at higher risk of contracting the virus. Vaccination ensures long-term protection and reduces the spread of the disease.Chickenpox is generally a mild disease, but it can lead to serious complications, especially in young children and pregnant women. Complications can include bacterial skin infections, pneumonia, brain inflammation, and even death in severe cases. The chickenpox vaccine is the best preventive measure to protect children and pregnant women from these risks.
Chickenpox can cause serious complications such as severe skin infections, pneumonia, and brain inflammation, particularly in pregnant women and children. Vaccination is highly recommended to protect vulnerable groups. The vaccine not only prevents the disease but also reduces the risk of complications like scarring and organ damage. It's best to vaccinate both children and pregnant women to avoid the spread and complications of chickenpox.
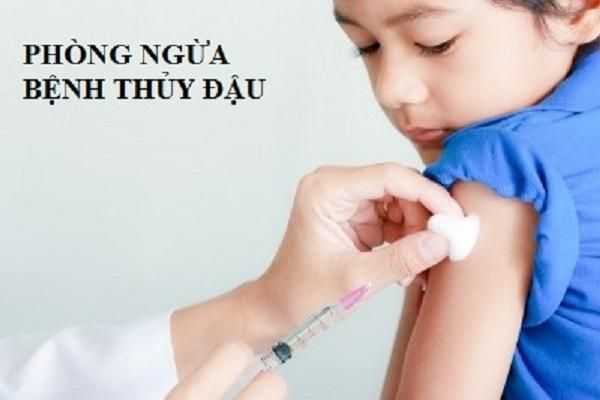 The chickenpox vaccine is given in two doses: the first dose at 12 months of age and the second dose between 4 and 6 years old. This vaccination schedule provides effective protection against the virus and minimizes the risk of severe complications associated with chickenpox.
The chickenpox vaccine is given in two doses: the first dose at 12 months of age and the second dose between 4 and 6 years old. This vaccination schedule provides effective protection against the virus and minimizes the risk of severe complications associated with chickenpox.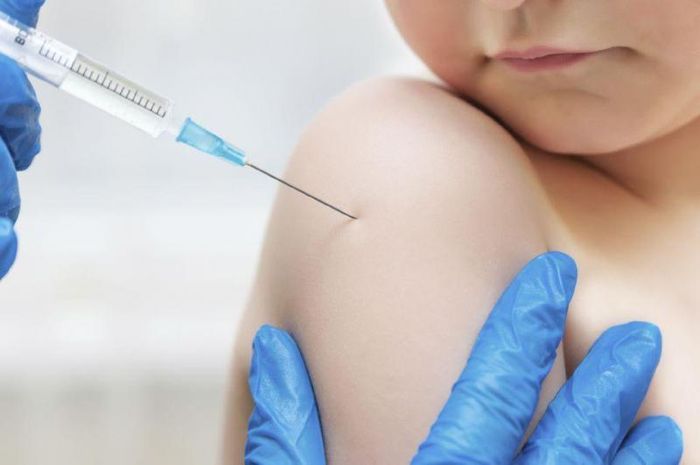 The chickenpox vaccine is a vital immunization to protect against the varicella virus, which causes the chickenpox infection. This vaccine helps prevent the spread of the disease and reduces the risk of complications, especially in young children and adults. Vaccinating against chickenpox is the most effective way to protect both individuals and communities from the virus.
The chickenpox vaccine is a vital immunization to protect against the varicella virus, which causes the chickenpox infection. This vaccine helps prevent the spread of the disease and reduces the risk of complications, especially in young children and adults. Vaccinating against chickenpox is the most effective way to protect both individuals and communities from the virus.Cervical cancer, often caused by the Human Papillomavirus (HPV), is a leading cancer affecting women between the ages of 15 and 44 in Vietnam. While not everyone infected with HPV will develop cervical cancer, over 90% of cervical cancer cases are linked to the virus. HPV infections can occur without symptoms and can lead to deadly consequences if left undetected. Vaccination against HPV is the most effective method to prevent cervical cancer, with a vaccine offering up to 90% protection for girls and women aged 9 to 26 years.
Cervical cancer is a significant health concern for women in Vietnam, with nearly 5,000 new cases diagnosed and over 2,000 deaths each year. The HPV vaccine is a critical preventive measure, helping to protect women from the virus that causes the majority of cervical cancer cases. It is recommended that girls and women between the ages of 9 and 26 receive the HPV vaccine, which provides long-lasting protection against the virus.
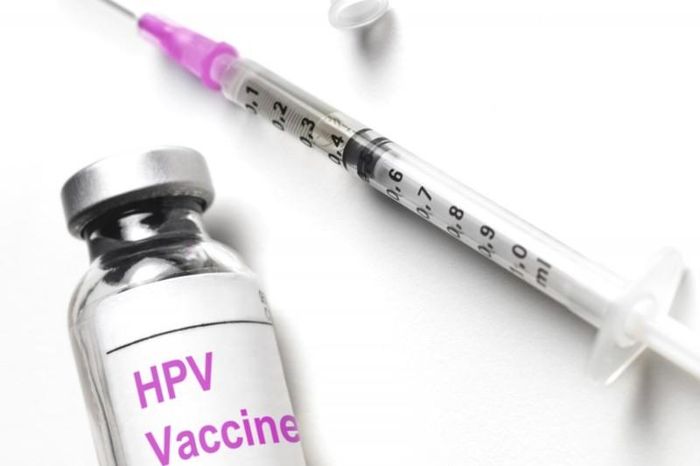 The HPV vaccine is administered in a three-dose schedule. The first dose is given on the designated vaccination day, followed by the second dose two months later, and the third dose six months after the first. This vaccination schedule ensures the best possible protection against the types of HPV responsible for cervical cancer.
The HPV vaccine is administered in a three-dose schedule. The first dose is given on the designated vaccination day, followed by the second dose two months later, and the third dose six months after the first. This vaccination schedule ensures the best possible protection against the types of HPV responsible for cervical cancer.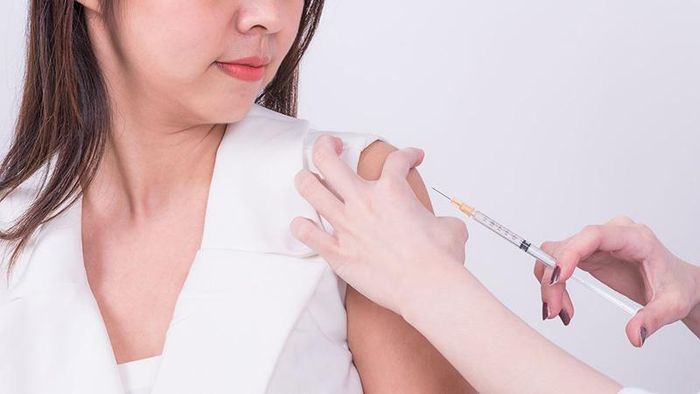 The HPV vaccine plays a critical role in preventing cervical cancer, which is one of the most common cancers among women. Human papillomavirus (HPV) is responsible for the majority of cervical cancer cases, and vaccination is the best defense. It is recommended for females between 9 and 26 years old to receive this vaccine to significantly reduce their risk of developing this cancer.
The HPV vaccine plays a critical role in preventing cervical cancer, which is one of the most common cancers among women. Human papillomavirus (HPV) is responsible for the majority of cervical cancer cases, and vaccination is the best defense. It is recommended for females between 9 and 26 years old to receive this vaccine to significantly reduce their risk of developing this cancer.Hepatitis B is a liver infection caused by the Hepatitis B virus, which spreads through blood, sexual contact, and from mother to child. Although it shares transmission routes with HIV, the Hepatitis B virus is 50-100 times more contagious. Vietnam has one of the highest rates of Hepatitis B infection worldwide, and vaccination remains the most effective preventive measure. However, the Hepatitis B vaccine does not protect against other types of hepatitis such as Hepatitis A or C.
The Hepatitis B virus is highly contagious, transmitted through blood, sexual activity, and from mother to child. It is far more transmissible than HIV, making it a serious concern, especially in countries like Vietnam. The most effective way to prevent Hepatitis B is through vaccination, though it does not provide immunity against other types of liver diseases. It's essential for those at risk to be vaccinated against this virus.
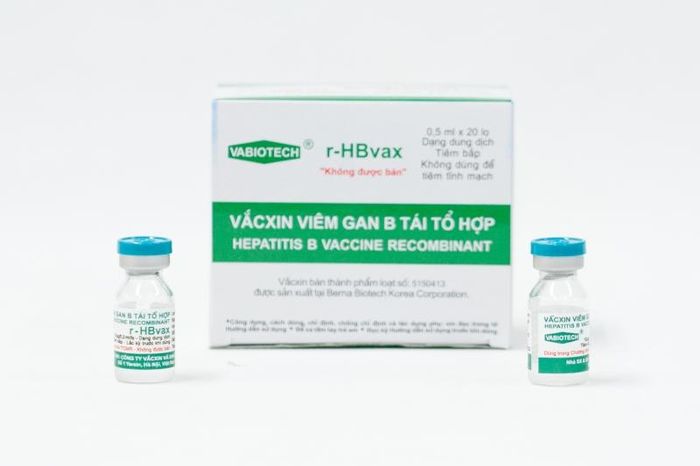 For children, the Hepatitis B vaccine is administered in a series of shots: the first dose should be given within 24 hours after birth. The second, third, and fourth doses can be combined with other vaccines (such as 6-in-1 or 5-in-1 vaccines) and should start when the child is 2 months old, with a minimum gap of 28 days between each shot. The final dose should be given when the child reaches 18 months, completing the vaccination schedule by 2 years of age.
For children, the Hepatitis B vaccine is administered in a series of shots: the first dose should be given within 24 hours after birth. The second, third, and fourth doses can be combined with other vaccines (such as 6-in-1 or 5-in-1 vaccines) and should start when the child is 2 months old, with a minimum gap of 28 days between each shot. The final dose should be given when the child reaches 18 months, completing the vaccination schedule by 2 years of age. Vacxin phòng viêm gan B
Vacxin phòng viêm gan B
Trước khi vắc-xin bạch hầu ho gà uốn ván (DTaP) được phát triển, ba căn bệnh này đã bùng phát thành dịch ở nhiều nơi trên thế giới. Bạch hầu là một bệnh ở đường hô hấp, có thể gây ra các vấn đề về hô hấp, tê liệt, suy tim và tử vong. Bệnh rất dễ lây lan khi người bệnh ho và hắt hơi. Uốn ván do một loại vi khuẩn thường được tìm thấy trong đất gây ra. Khi xâm nhập vào cơ thể, vi khuẩn sẽ tiết ra chất độc tấn công hệ thần kinh, gây co thắt cơ và tử vong nếu không được điều trị kịp thời. Ho gà gây ho và co thắt cổ họng nghiêm trọng, khiến trẻ sơ sinh gặp khó khăn khi ăn uống hoặc thậm chí là thở. Bệnh cũng rất dễ lây lan, có thể dẫn đến viêm phổi, co giật, tổn thương não và tử vong. Vắc-xin bảo vệ cộng đồng bằng cách ngăn chặn sự lây lan từ người này sang người khác, ngoài ra còn là cách hạn chế mắc bệnh cho người chưa được tiêm chủng. Nếu mọi người ngừng tiêm vắc-xin, tỷ lệ mắc ba căn bệnh này sẽ nhanh chóng tăng lên, hàng ngàn bệnh nhân mới sẽ xuất hiện và có nguy cơ tử vong cao.
Chống chỉ định:
- Phản ứng dị ứng trầm trọng (ví dụ, chứng quá mẫn) sau khi dùng liều trước hoặc với thành phần của vắc xin.
- Đối với thành phần ho gà: Bệnh não (như hôn mê, suy giảm nhận thức, co giật kéo dài) xảy ra trong vòng 7 ngày sau khi dùng DTaP hoặc Tdap trước và điều này không phải do nguyên nhân khác.
Lộ trình tiêm vắc-xin phòng DTaP cho trẻ em:
- Một liều lúc 2 tháng tuổi.
- Một liều lúc 3 tháng tuổi.
- Một liều lúc 4 tháng tuổi.
- Một liều lúc 18 - 24 tháng tuổi.
- Một liều lúc 4 - 6 tuổi.
 Vacxin phòng DTaP
Vacxin phòng DTaP DTaP Vaccine
DTaP Vaccine
12. Haemophilus Influenza Type B (Hib) Vaccine
Haemophilus influenzae type B (HIB) bacteria are recognized as one of the leading causes of bacterial meningitis in infants and young children. Since their immune systems are still developing, if not treated promptly, these children may face severe long-term consequences such as cognitive impairments, hearing loss, and neurological damage. The fatality rate for bacterial meningitis is between 5% and 10%. HIB bacteria are commonly found in the nasal passages and throat, making it highly contagious through airborne transmission. This results in rapid outbreaks within communities. Many children carry the bacteria without showing symptoms, but infants and toddlers aged 6 months to 2 years are especially vulnerable. The good news is that this disease can be effectively prevented through vaccination.
Contraindications:
- Severe allergic reactions (such as anaphylaxis) following a previous dose or due to any component of the vaccine.
- Any form of illness, mild or severe, with or without fever (vaccination should be delayed until recovery).
Recommended Hib Vaccine Schedule:
- The first dose should be administered between 2 to 4 months of age.
- A booster dose should be given between 16 to 18 months of age.
 Haemophilus Influenza Type B (Hib) Vaccine
Haemophilus Influenza Type B (Hib) Vaccine Haemophilus Influenza Type B (Hib) Vaccine
Haemophilus Influenza Type B (Hib) Vaccine























